Our vision for research transparency is that trusted information from health and social care research studies is publicly available for the benefit of all.
All research should be registered in a publicly accessible database. For clinical trials, it is a condition of a favourable ethics opinion. Following a favourable opinion from a Research Ethics Committee (REC), the sponsor should register the study on a World Health Organisation (WHO) compliant registry before the first participant is recruited, and no later than 6 weeks after.
It is recommended that researchers use either the UK-based registry ISRCTN or the US-based registry ClinicalTrials.gov. These registries feed in to ‘Be Part of Research', an online service run by the National Institute for Health and Care Research (NIHR), which helps members of the public understand what research is, what it might mean to take part, and shows what research is currently happening across the UK.
In 2023, we published, for the first time, a full list of clinical trials that had received a favourable opinion from a REC in 2022, with the registration details that we hold for them.
Last year, as part of our commitment to publish transparency performance in the research community, we published a full list of clinical trials that received a favourable opinion from a REC in 2023, with the registration details we hold for them.
We are repeating this again this year for studies which received a favourable opinion from the REC in 2024. The full list is available as a CSV file.
We are also publishing a table of clinical trials which were given a favourable opinion from a REC in 2024 which we have not received a registration number for. The table contains data we held in January 2026. Updates after this date will be appear on the research summaries webpage. This one remains a snapshot of the position this week.
What we are publishing
We are sharing data on the registration status of clinical trials in the UK that received a favourable opinion from RECs in 2024, including their registration number or an explanation for non-registration where this is known.
Clinical trials are defined as:
- clinical trial of an investigational medicinal product
- clinical investigation or other study of a medical device
- combined trial of an investigational medicinal product and an investigational medical device
- other clinical trial to study a novel intervention or randomised clinical trial to compare interventions in clinical practice
Please note that in our data published values have been rounded up and may not add up to 100%.
What we found
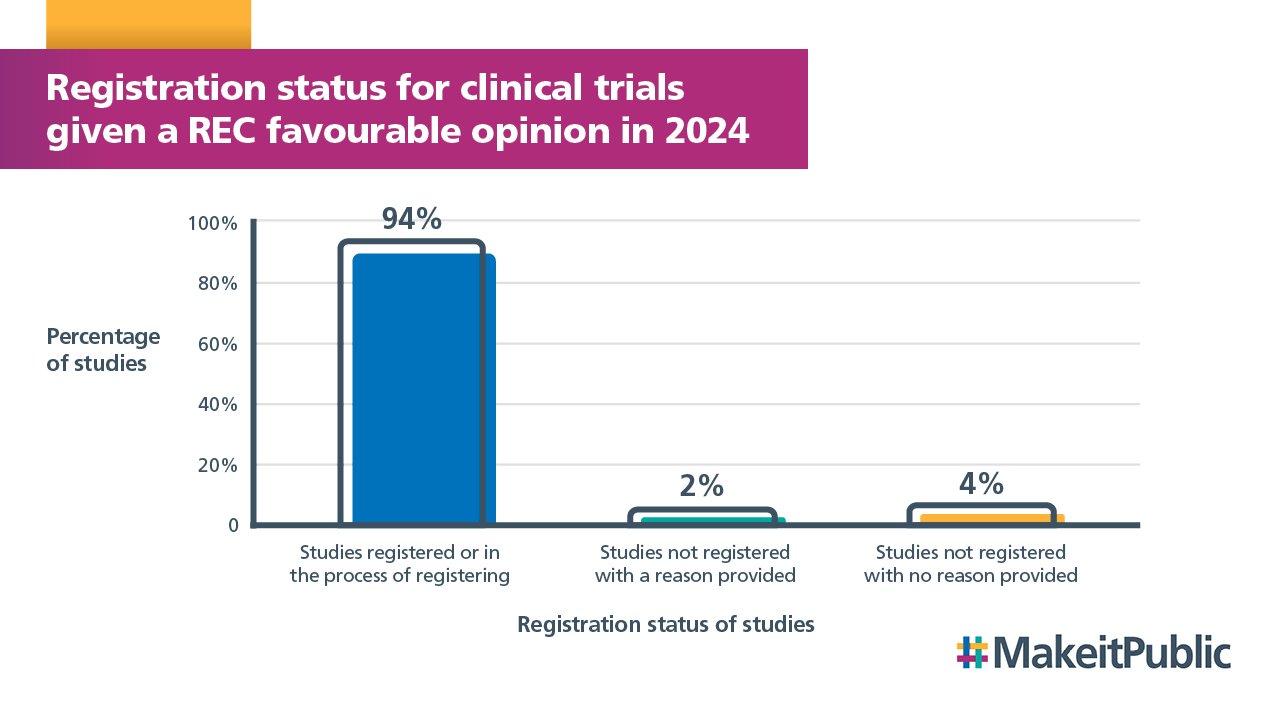
At the time of publication of this report, 94% of clinical trials that received a favourable opinion from a UK REC in 2024 have provided the HRA with a registration number, with a further 2% in the process of registering.
4% of clinical trials have not provided the HRA with a registration number and the sponsor has not told us why.
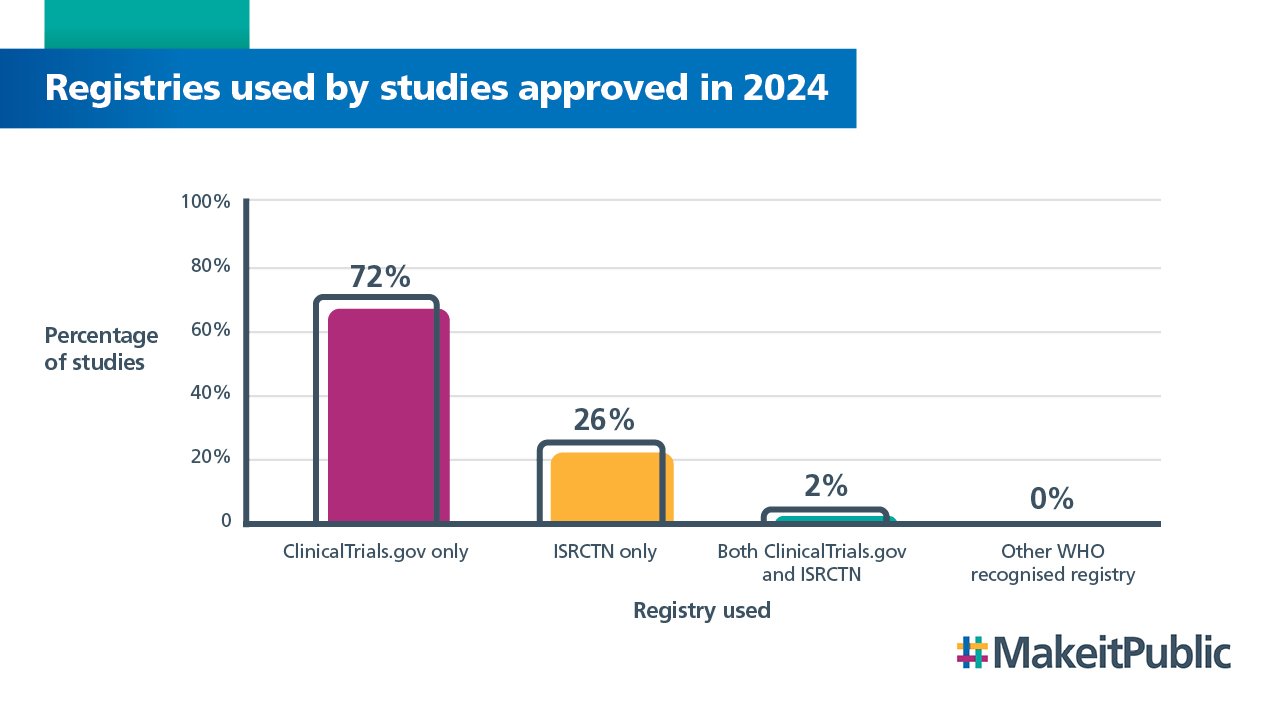
The most popular registry used for clinical trials given a favourable opinion in 2024 was ClinicalTrials.gov, which is the same as in 2022 and 2023.
The rates are the same as last year’s report, except for a 1% increase in trials registered on ISRCTN.
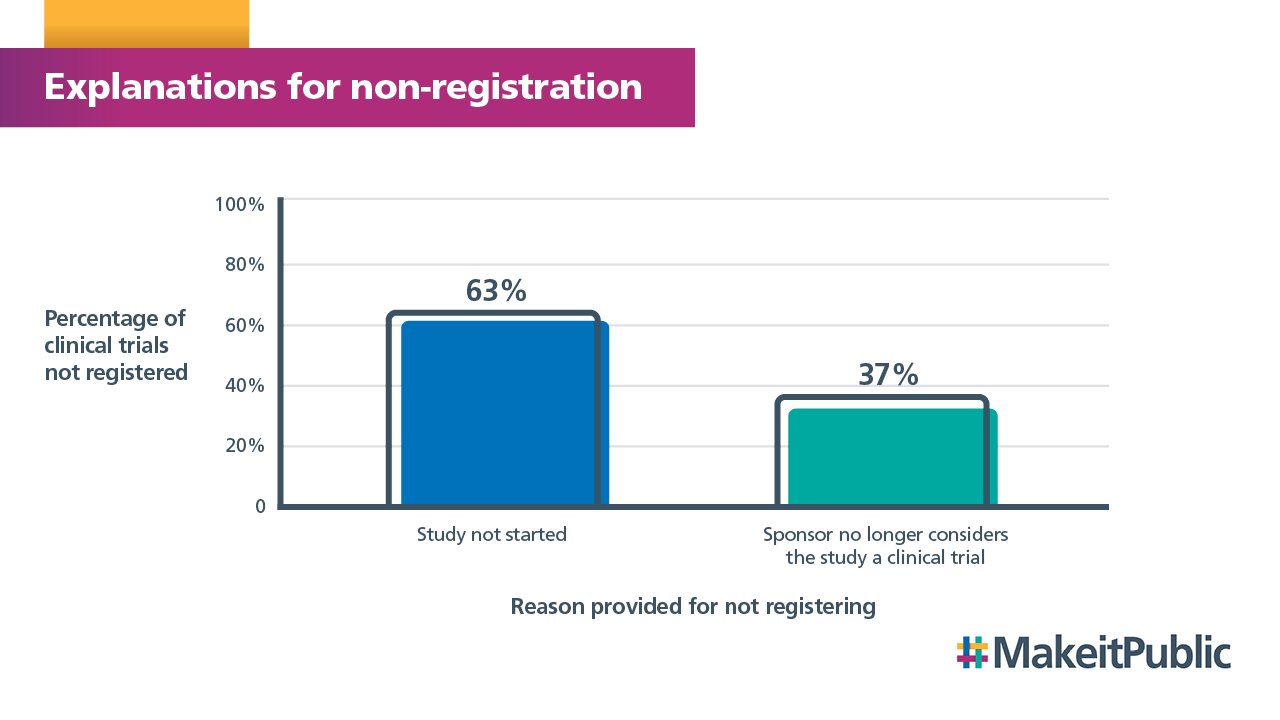
Of the 4% who have provided a reason for non-registration, the most common explanation was that the study had not started because recruitment had not yet begun or the study had been withdrawn. There were also a significant number of sponsors who no longer considered their study to be a clinical trial.
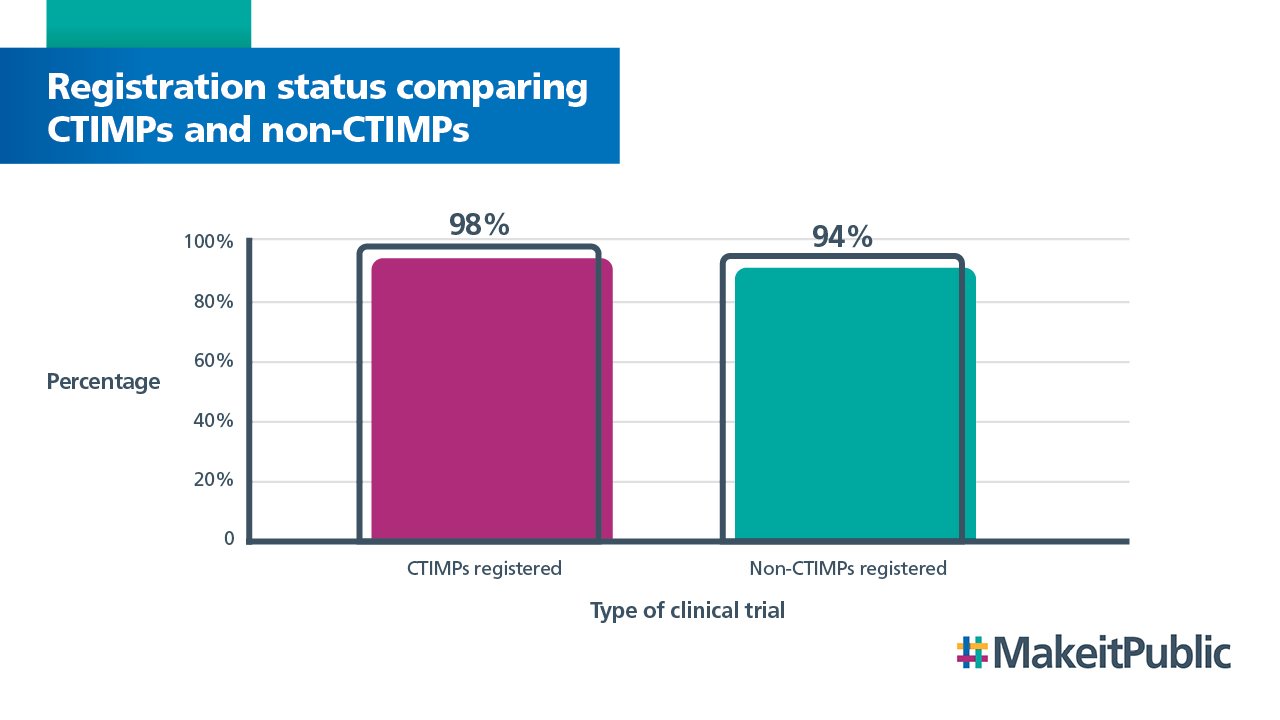
We also compared the registration status of clinical trials of an investigational medicinal products (CTIMPs) and non-CTIMPs. This show that registration compliance is higher for CTIMPs than non-CTIMPs. Compliance for both study types has increased since last year.
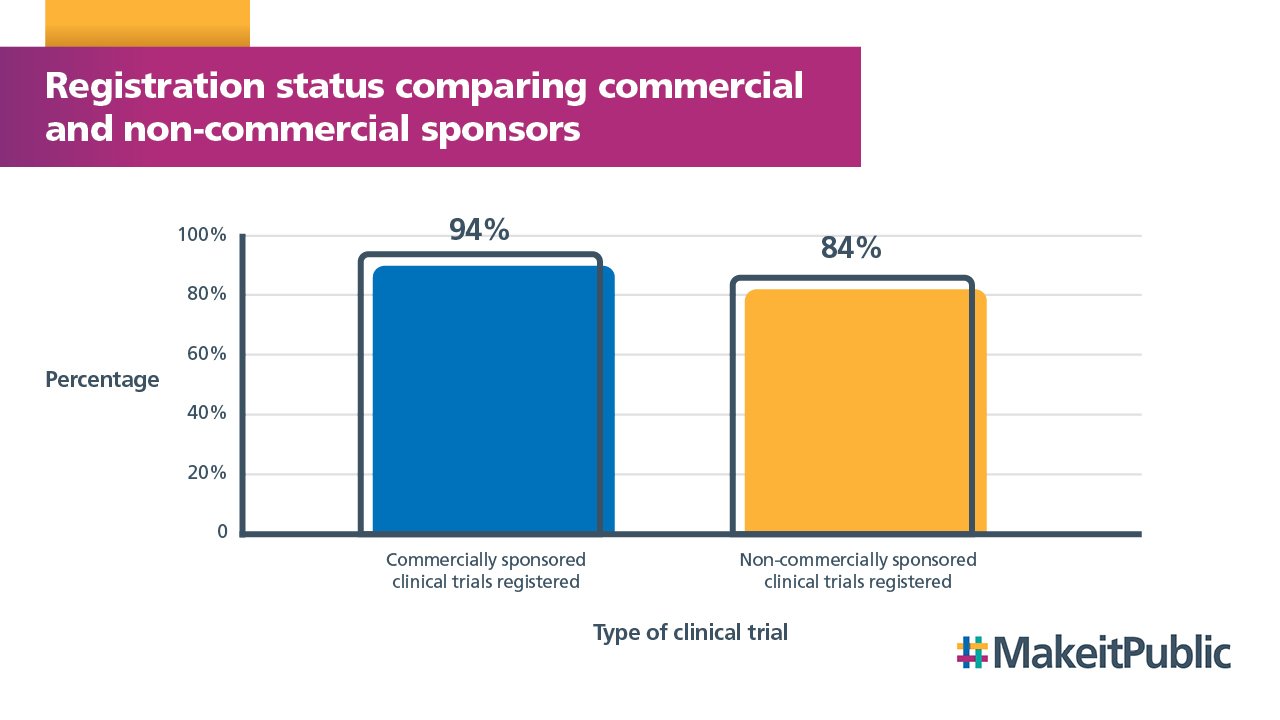
We also compared registration status for commercial and non-commercial studies. This found that registration compliance is higher for commercially sponsored clinical trials than non-commercially sponsored trials.
Registration data
The below table sets out clinical trials which were given a favourable opinion from a REC in 2024, which we have not received a registration number for.
Where a reason for non-registration was provided at the point the study was contacted 12 months after receiving REC approval, this has been included.
If your study is on this list and you would like to inform us of the registration number please email us at study.registration@hra.nhs.uk and this will appear on the research summaries webpage.
The studies are arranged in order of IRAS number. This is a number given to a study when the first application for approvals is made and which follows the study through its lifecycle, until the results are published.
The full registration data set of clinical trials that received a favourable opinion from a REC in 2024 is available as a CSV file.
There is not one single registry for clinical trials. The format of the number in the csv.file can tell you which register the study has used.
ISRCTN - Numbers beginning ISRCTN
Clinicaltrials.gov - Numbers beginning NCT
ANZCTR - Numbers beginning ACTRN
DRKS - Numbers beginning DRKS
| IRAS project ID | Full study title | Sponsor organisation | Reason for non-registration |
| 246287 |
Proof of concept study of the effect of hand exercise and ischaemic preconditioning on vascular function in CKD patients |
East and North Hertfordshire NHS trust |
Registration currently underway |
| 264457 |
Impact of urodynamic and video-urodynamic testing on surgical outcomes in women with recurrent urinary incontinence - A feasibility study |
University College London | |
| 270562 |
Nutritional screening and intervention in patients undergoing treatment for cardiovascular disease. |
University of Hull Teaching Hospitals Trust |
|
| 287197 |
Evaluating the feasibility and perceived acceptability of a novel guided internet-based therapy for Complex Post-Traumatic Stress Disorder– Pilot Study and Randomised Controlled Trial (ENABLE) |
Cardiff University |
Sponsor no longer considers this a clinical trial |
| 288916 |
Pilot study: Structural Magnetic Resonance Imaging of the Neonatal Lung |
Sheffield Teaching Hospital NHS Foundation Trust |
Sponsor no longer considers this a clinical trial |
| 297176 |
Exploring the potential for 18F-Fluoro-ethyl-tyrosine (18F-FET) to enable wider access to molecular imaging for patients with pituitary adenomas (FET-pit-PET study) |
Cambridge University Hospitals NHS Foundation Trust & The University of Cambridge |
|
| 301613 |
Improving Pelvic Rehabilitation using Epidural Stimulation after SCI |
University College London | |
| 302325 |
VMAT Radiotherapy for the Emergency Management of Metastatic Spinal Cord Compression |
The Newcastle upon Tyne Hospitals NHS Foundation Trust |
|
| 311461 |
Single-centre pilot-study to determine the optimal sacral neuromodulation (SNM) stimulation parameters to treat idiopathic detrusor overactivity and dysfunctional voiding. |
Guy's and St Thomas' Foundation NHS Trust | |
| 314080 |
Regenerative Potential of Tissue Specific Cell-Based Therapies for Alveolar Ridge Preservation. A Randomised Controlled Clinical Trial |
University College London | |
| 316052 |
InnoVative ObservAtionaL In-vitro DiAgnosTic pErformance study (VALIDATE). A complex innovative design study to enable the development assessment and analysis of evidence required to establish or verify the scientific analytic and observational clinical validity of in vitro diagnostics |
Manchester University NHS Foundation Trust |
Recruitment not yet started |
| 318799 |
The effectiveness of Otinova (Burow's solution) for mild-moderate Acute Otitis Externa in primary and secondary care: A feasibility study |
Cambridge University Hospitals NHS Foundation Trust & The University of Cambridge |
|
| 320918 |
Evaluation of structure and vascularity of glaucoma surgical sites using Optical Coherence Tomography (The GHOST Study) |
University of Liverpool | Registration currently underway |
| 323606 |
Feasibility of a randomised controlled trial (RCT) to examine the effectiveness and cost-effectiveness of auditory-cognitive training to improve outcomes for NHS patients who are first-time hearing aid users compared with hearing aids alone |
Nottingham University Hospitals NHS Trust | Recruitment not yet started |
| 323764 |
Reliability of self-reported smoking status against exhaled Carbon Monoxide (eCO) measurement in patients undergoing fusion surgery around the foot and ankle |
Golden Jubilee Foundation | Recruitment not yet started |
| 324487 |
A randomised controlled study to explore the safety and efficacy of the mySmartCOPD treatment re-alignment algorithm as part of a digital self-management app in patients with COPD |
University of Southampton | Recruitment not yet started |
| 324564 |
Modulation of endothelial function in Alzheimer's Disease by 18kDa Translocator Protein |
Imperial College London | Recruitment not yet started |
| 324891 |
BREATHE feasibility study (Biofeedback Reactivity and Awareness To HEalth): The effect of biofeedback and mindfulness breathing on blood pressure and psychological wellbeing |
University Hospitals of Leicester NHS Trust |
Registration currently underway |
| 325353 |
Can an eSexual Health Clinic improve health outcomes for people with chlamydia and their partners? The SEQUENCE Digital cluster crossover randomised controlled trial. |
Central and North West London NHS Foundation Trust |
Recruitment not yet started |
| 325793 |
Prospective observational study to evaluate safety and performance of Spacer-K ATS in the PJI of the knee joint |
Tecres S.p.A | Recruitment not yet started |
| 326211 |
Assessment of response to furosemide therapy by bioelectrical impedance versus cumulative fluid balance recording |
Great Ormond Street Hospital for Children NHS Foundation Trust |
Registration currently underway |
| 326227 |
pErsonalised Nocebo Assessment of Beta-bLockEr symptoms in Heart Failure. Are the adverse side effects of beta-blockade in heart failure genuine? An n-of-1 personalised assessment |
Imperial College London & Imperial College Healthcare NHS Trust |
|
| 326983 |
Sodium-glucose Transporter Type 2 Inhibition in Anthracycline-related Cardiotoxicity |
University of Aberdeen | Recruitment not yet started |
| 327077 |
Pulsed Field Ablation during Left Atrial Appendage Occlusion: A Randomised Controlled Trial |
Liverpool Heart & Chest Hospital NHS Foundation Trust |
Recruitment not yet started |
| 327103 |
Pilot study for rapid pathology techniques on surplus free flap tissue from head and neck cancer reconstructive surgery |
University College London Hospital NHS Trust |
Registration currently underway |
| 327122 |
VITROS Immunodiagnostic Products Progesterone 2 Assay Apparently Healthy Sample Collection |
QuidelOrtho Inc | |
| 327368 |
Comparing pOrtable MRI and TRUS-Fusion Prostate biopsy to Assess aCcuracy of prostate cancer detection (IP10-COMPACT) |
Imperial College London | Recruitment not yet started |
| 327879 |
Cerclage after Caesarean: a randomised controlled trial to assess the optimal preventative management for preterm birth secondary to caesarean section damage (ABOVE) |
King's College London | |
| 328297 |
Ablation of Focal Activation During Persistent Atrial Fibrillation to Determine the Characteristics of Focal Drivers |
Imperial College London | |
| 329260 |
Feasibility Acceptability Safety and Preliminary Effectiveness of an Ultra-Brief Crisis IPT-A Based Intervention (IPT-A-SCI) and Treatment as Usual (TAU) for Suicidal Children and Adolescents in UK CAMHS |
NHS Lanarkshire |
Sponsor no longer considers this a clinical trial |
| 329343 |
Evaluation of Synvichor in patients with undifferentiated joint pain to discriminate between infectious and non-infectious joint inflammation |
OrthoDx |
Sponsor no longer considers this a clinical trial |
| 329409 |
A multicentre randomised trial of surgical versus percutaneous revascularisation of ischaemic left ventricular dysfunction (iLVSD) in the United Kingdom with embedded internal pilot and health economic analysis |
University of Leicester | |
| 329602 |
Catheter Ablation versus Medical Rate Control in Atrial Fibrillation with Systolic Heart Failure and Myocardial Fibrosis – an MRI guided Multi-Centre Randomised Controlled Trial |
Alfred Hospital | |
| 330287 |
The WISH Trial (Web-based versus standard Information for Same day Hysterectomy): An evaluation of an on-line patient information resource for same day discharge following laparoscopic hysterectomy (SDD-LH) |
Birmingham Women`s and Children`s NHS Foundation Trust |
|
| 330364 |
Reliability and feasibility study of electronic wheeze detection device (WheezeScan) in children with recurrent or a recent wheeze attack |
Imperial College London |
Sponsor no longer considers this a clinical trial |
| 330578 |
Cogigame Clinical Study: Feasibility Study and Randomised Controlled Trial of Combined Electroencephalogram (EEG) and Virtual Reality (VR) for Upper-Limb Rehabilitation in Post-Stroke Patients |
Cogitat | |
| 330916 |
The use of Entropy to support diagnoses in sleep disordered breathing |
University College London | Recruitment not yet started |
| 330988 |
Neurofeedback Treatment for Nociplastic Pain in Rheumatoid Arthritis |
NHS Greater Glasgow & Clyde | |
| 331944 |
TECTONIC: Transnasal Endoscopy versus ConvenTional endOscopy for the diagNosIs of Coeliac disease in adults: a comparison of accuracy and tolerability |
Sheffield Teaching Hospital NHS Foundation Trust |
|
| 332335 |
Chair based rehabilitation for massive irreparable rotator cuff tears. A pilot study |
Canterbury Christ Church University | |
| 332374 |
Feasibility and Acceptability of a Home-Based Adaptable Telerehabilitation Programme for Transcatheter Aortic Valve Implantation (TAVI) Patients. |
Northumbria University |
Sponsor no longer considers this a clinical trial |
| 332448 |
Remote Acute Monitoring-based Same-day Discharge Strategy of Post Transcatheter Aortic Valve Implantation Patients Versus Standard Care at 30-Day Follow-up (TELE-TAVI Trial) |
Imperial College London | |
| 332594 |
Human BronchiectAsis RhinoviRus challenge to define Immunopathogenesis of ExaceRbation |
Imperial College London |
Sponsor no longer considers this a clinical trial |
| 332706 |
What effect does looking at red light have on the eye in children and young people? – A proof-of-concept study |
University College London |
Sponsor no longer considers this a clinical trial |
| 332905 |
Improving Uptake of Bowel Cancer Screening Among South Asian Adults in Bradford |
University of Hull | |
| 333306 |
The feasibility of an exercise based digital self-management platform for people living with lung cancer to improve physical functioning and well being |
Oxford Brookes University |
Sponsor no longer considers this a clinical trial |
| 333382 |
Developing a handheld anterior segment optical coherence tomography device to assess Kayser Fleischer rings in Wilson's disease |
Royal surrey NHS Foundation Trust | |
| 333900 |
Investigating the use of TRANScutaneous vagus nerve stimuLATion for the treatment of strokE related aphasia (TRANSLATE): a pilot RCT |
Sheffield Teaching Hospital NHS Foundation Trust |
|
| 334071 |
Accuracy and repeatability of the Tono-Vera rebound tonometer |
The Queen's Medical Research Institute | |
| 334426 |
Physiological changes during chemoradiotherapy for HPV-related oropharyngeal squamous cell carcinoma |
The Newcastle upon Tyne Hospitals NHS Foundation Trust |
Sponsor no longer considers this a clinical trial |
| 334551 |
Carer Involvement in exercise programmes for acute Neurosciences patients with acquired brain injury |
Norfolk and Norwich University Hospital NHS Foundations Trust |
|
| 334672 |
Arts for the Blues for Children: Intervention Development |
Alder Hey Children's NHS Foundation Trust |
Sponsor no longer considers this a clinical trial |
| 334773 |
An experimental medicine study to investigate the role of the 18 kiloDalton Translocator Protein in glucose metabolism |
Imperial College London | Recruitment not yet started |
| 334935 |
Safety of Early discharge Using index microcirculatory Resistance in patients with acute myocardial infarction |
Newcastle upon Tyne Hospitals NHS Foundation Trust |
|
| 334973 |
A post-market clinical follow-up (PMCF) study to evaluate clinical radiographic and patient reported outcomes following a hip hemi-arthroplasty using Corin devices. |
Corin Ltd | Recruitment not yet started |
| 335176 |
Determining reference ranges for a novel Light Emitting Diode (LED) phototest device |
University of Dundee | Registration currently underway |
| 335406 |
Evaluating a digital application designed to support young people with gastrointestinal functional disorders: a feasibility study |
University of Aberdeen and NHS Grampian | Recruitment not yet started |
| 335504 |
A Dyadic approach to Acceptance and Commitment Therapy (ACT) for people living with dementia and their caregivers |
Univeristy of Lincoln | Recruitment not yet started |
| 335566 |
Prospective non-randomised single arm study on visual and optical outcomes after mini-monovision with hydrophobic monofocal intraocular lens |
University Hospitals Sussex NHS Foundation Trust |
|
| 335739 |
A qualitative exploration of the acceptability and experiences of music therapy sessions for persons with dementia and their carers living at home |
Cumbria Northumberland Tyne and Wear NHS Trust |
Sponsor no longer considers this a clinical trial |
| 335814 |
A single blinded randomised trial into the efficacy of silicone sheeting for cutaneous scars. The Best Evidence for Scar Treatment (BEST) 1 group trial. |
Buckinghamshire Healthcare NHS Trust | Recruitment not yet started |
| 336063 |
Interferential therapy for the treatment of children with gastrointestinal dystonia; an acceptability study |
Birmingham Women`s and Children`s NHS Foundation Trust |
|
| 336290 |
Non-invasive Monitoring of Glucose in Diabetes Mellitus: A Pilot Study to Evaluate Photonic Chips as a Non-Invasive Glucose Sensor in Type 1 Diabetics in a Laboratory Prototype Device |
University of Leeds | Registration currently underway |
| 336429 |
JIA (Juvenile Idiopathic Arthritis) Toolbox Feasibility Study |
Sheffield Children's NHS Foundation Trust | |
| 336506 |
TinnSpire: Open-label Single Arm non-randomised Feasibility study of Bimodal Transcutaneous auricular vagus nerve stimulation & Broadband Shaped-Noise Sound Therapy with Slow Resonant Breathing for the treatment of tinnitus |
University of Sussex | |
| 336523 |
Holistic AppRoach for Multimorbidity OptimisatioN after IntenSivE care (HARMONISE) |
Queen's University Belfast | Recruitment not yet started |
| 336699 |
A feasibility study to explore rehabilitation outcomes Usability and User Experience of a Web-Based Platform for delivering treatment for cognitive and language impairment amongst adults with acquired neurological disorders |
Liverpool University Hospitals NHS Foundation Trust |
Registration currently underway |
| 337141 |
An observational, registry-based study of performance and safety data from participants treated for open angle glaucoma or ocular hypertension with the Eagle® device |
Belkin Vision Ltd | |
| 337192 | Artix Validation Study | Ossur | Recruitment not yet started |
| 337305 |
MR imaging only radiotherapy planning site-specific analysis: a prospective technical validation and clinical feasibility study (MIMOSA) |
The Newcastle upon Tyne Hospitals NHS Foundation Trust |
Recruitment not yet started |
| 337472 |
Magnesium deficiency as a reversible driver of vascular complications in type 1 diabetes |
University of Leeds | |
| 338078 |
Acceptability (including gastrointestinal tolerance and compliance) of a plant based high energy paediatric enteral tube feed. |
Nestle Health Sciences | |
| 338473 |
A Pilot Study of Cognitive Remediation Therapy for Autistic Adolescents |
King's College London | Registration currently underway |
| 338543 |
A randomised controlled pilot study of the effectiveness of a Virtual Reality headset on post-operative Relaxation Anxiety and Pain in patients undergoing elective laparoscopic colorectal surgery |
East Kent Hospitals Universities Foundation Trust |
Registration currently underway |
| 338881 |
A Pilot Study of 2RT Laser Application in Intermediate Age-Related Macular Degeneration (AMD) |
Nova Eye Medical | Recruitment not yet started |
| 339260 |
The use of Hemodynamic Occlusive Vascular Response (HOVRâ„¢) technology to assess the impact of peripheral artery disease (PAD) on tissue perfusion and oxygenation in the foot |
VOTIS Subdermal Imaging Technologies LTD | Recruitment not yet started |
| 339361 |
Visual and stereoacuity: average paraxial power and spherical equivalent |
Royal Liverpool University Hospital | |
| 339595 |
cReative psychological Evidence-bAsed intervention to support the wellbeing of doctors-in-Training – fEasibility study |
Pennine Care NHS Foundation Trust |
Sponsor no longer considers this a clinical trial |
| 339632 |
Intraoperative use of spectral imaging and Laser Speckle to detect colorectal cancer and provide quality assurance during surgery |
University College London Hospital NHS Trust |
Registration currently underway |
| 339642 |
A feasibility trial of a skills enhancing programme for carers of people presenting with complex emotional needs and/or chronic emotion dysregulation – Carers All Require Emotional support, Resilience and Skills: The CARERS Trial |
University of York | |
| 339771 |
The Effectivness Feasibility and Acceptability of Virtual Reality Therapy Integrated Into Cognitive Behavioural Therapy For Emetophobia: A Single Case Experimental Design |
South London & Maudsley NHS Foundation Trust |
Sponsor no longer considers this a clinical trial |
| 339786 |
An Online Acceptance and Commitment Therapy Intervention for Adults with Hereditary Angioedema: A Feasibility Study |
University of Surrey | |
| 339966 |
A prospective randomised controlled trial (RCT) to assess the impact and benefits of using an AI system in live double reading practice for breast cancer screening |
Kheiron Medical Technologies Ltd | Recruitment not yet started |
| 340016 |
Exploring the effect of a novel psychological intervention for negative symptoms based on transdiagnostic mechanisms of change |
University of Glasgow |
Sponsor no longer considers this a clinical trial |
| 340260 |
Endoscopic submucosal resection vs. endoscopic full thickness resection for the treatment of rectal neuroendocrine tumours from 10 to 20 mm: a multi-centre randomised trial |
Royal Free NHS Foundation Trust | Recruitment not yet started |
| 340693 |
PROMOTE: A double-blinded randomised placebo-controlled parallel trial to assess the impact of prebiotic supplementation on the wellbeing of young adults with self-reported mild or moderate stress. |
University of Southampton | Recruitment not yet started |
| 340847 |
Pilot study to develop machine learning algorithms for the pre-symptomatic detection of deterioration in patients undergoing chimeric antigen receptor T-cell therapy |
University College London Hospital NHS Trust |
Registration currently underway |
| 340890 |
Personalised process-based psychological intervention for paediatric headache cases: An idiographic ecological momentary study with weekly feedback provision |
Royal Holloway University of London | Registration currently underway |
| 341164 |
Acceptability (including gastrointestinal tolerance compliance and palatability) of a paediatric oral nutritional supplement containing food blends |
Nestle Health Sciences | Registration currently underway |
| 341268 |
A prospective dual center double armed open label clinical Study to Evaluate the Performance of the PathKeeper System for Pedicle Screw Positioning in spine surgery. |
PathKeeper Surgical Ltd. | |
| 341778 |
Clinical utility of ctDNA in detecting resistance mechanisms and delivering precision medicine: a tumour agnostic study |
The Royal Marsden NHS Foundation Trust | Registration currently underway |
| 341827 |
Feasibility testing of an intervention aimed at improving cancer investigation referrals in general practice for patients with anxiety and/or depression |
University of Exeter |
Sponsor no longer considers this a clinical trial |
| 342058 |
N-WATCH: Nottingham Wheeze Assessment in Tidal Children - Harnessing Capnograms for Airway Health – Feasibility Observational Study |
University of Nottingham | Registration currently underway |
| 342215 |
Portable imaging solutions towards affordable healthcare: A feasibility study for microfluidic device for the detection of disease specific proteins |
Medway NHS Foundation Trust | Recruitment not yet started |
| 342657 |
Investigating the effect of the combination of L-carnitine tartrate dihydrocapsiate and protein yeast hydrolysate (DNF10) for 12 weeks on energy intake and energy expenditure in overweight and obese adults: A Randomised Controlled Trial |
Societie des Produits Nestle S.A. | |
| 342936 |
Feasibility of an online Compassion Focused Therapy (CFT) intervention for children and young people (CYP) aged 14-18 with Type 1 Diabetes. |
University of Surrey | |
| 343924 |
The psychological impact of eating sourdough bread compared to industrial store-bought bread: a single case experimental design (SCED). |
Royal Holloway University of London | Recruitment not yet started |
| 344071 |
Evaluating novel OPM-magnetoencephalography in pre-surgical mapping for patients with epilepsy |
Aston University | Registration currently underway |
| 344154 |
Post Market Clinical Follow Up and Continuous Development of Upper Limb Prosthetic Devices |
Ossur | Recruitment not yet started |
| 344310 |
Clinical Performance Study Protocol for Use of VENTANA PD-L1(SP263) CDx Assay in GSK Study 213823 (GALAXIES LUNG-301) |
Ventana Medical Systems | |
| 345553 |
Assessing Treat-to-Target Feasibility and Biomarker Monitoring in Childhood Systemic Lupus Erythematosus: The NEPHROTARGET Study |
University of Liverpool |
Sponsor no longer considers this a clinical trial |
| 345942 |
Gluten-Free Refractory CBT: an online Cognitive Behaviour Therapy (CBT) group intervention designed to reduce anxiety in adults with refractory coeliac disease |
University of Surrey |
Sponsor no longer considers this a clinical trial |
| 345962 |
Positive Mental Training for sleep and mood outcomes: A single case experimental design |
University of Edinburgh |
Sponsor no longer considers this a clinical trial |
| 345992 |
A Feasibility Study Employing a Randomised Controlled Trial to Compare the Effectiveness of VR-Assisted Nature Therapy with an Established Online Intervention for Stress Management |
EarthScape VR Limited | |
| 346033 |
AAV8 TAb Assay for Eligibility in the R131L1265-HEMB-2318 Clinical Trial |
ARUP Laboratories |
Registration not required as this refers to a companion device for a CTIMP that is registered |
| 346045 |
A Pivotal Single-Arm Single-Centre Clinical Investigation to Evaluate the Safety and Efficacy of the Sentire Surgical System (C1000) in Robotic-Assisted Abdominal Soft Tissue Surgery |
Cornerstone Robotics Limited | Registration currently underway |
| 346124 |
The effect of three hours of uninterrupted sitting on central and peripheral arterial stiffness in Long COVID |
University of Winchester | Recruitment not yet started |
| 346250 |
Early help for parents and carers of adolescents who self-harm: a pilot feasibility randomised control trial of a Dialectical Behaviour Therapy-informed workshop. |
King's College London | Registration currently underway |
| 347008 |
Mentalisation and Group Cohesion: An Investigation into the Mechanisms and Moderators of Change within CBASP-G. |
University of Edinburgh |
Sponsor no longer considers this a clinical trial |
| 347547 |
Clinical validation of the PneumoRator respiratory rate sensor in a perioperative cohort |
University Hospital Southampton NHS Foundation Trust |
Registration currently underway |
| 347639 |
System Usability of the MedTracks App to aid to Medication Adherence (SBRI) |
NHS Highland | Registration currently underway |
| 347696 |
Effects of FES arm cycling on functional ability and strength in people with spinal cord injury: A feasibility study |
Glasgow Caledonian University | Registration currently underway |
| 348079 |
Performance of the iFAST System for Rapid Phenotypic Antimicrobial Susceptibility Testing (AST) of Gram Negative Bacteria directly from Positive Blood Culture Bottles and directly from growth isolated colonies |
iFAST Diagnostics Limited |
Sponsor no longer considers this a clinical trial |
| 351043 |
Motor Neuron Disease (MND) Alert: a mixed-method feasibility and acceptability pilot study |
University of Sheffield | Registration currently underway |
| 1004917 |
A Phase 2 randomized double-blind placebo-controlled parallel group study (TRANSFORM) to evaluate the efficacy and safety of GSK3915393 in participants with Idiopathic Pulmonary Fibrosis (IPF) |
GlaxoSmithKline Research & Development Limited |
|
| 1007729 |
Evaluating the effects of GLP-1 analog as a treatment for sporadic behavioral variant frontotemporal dementia (bvFTD) |
Imperial College London & Imperial College Healthcare NHS Trust |
|
| 1007861 |
A Phase 1/2 Multicenter Open Label Dose Escalation & Dose Expansion Study of JK08 an IL-15 Antibody Fusion Protein Targeting CTLA-4 Monotherapy or in Combination in Patients with Unresectable Locally Advanced or Metastatic Cancer |
Salubris Biotherapeutics Inc. | Recruitment not yet started |
| 1009088 |
A Randomised Double-blind Placebo-controlled Parallel-group Phase 2A Trial to Assess the Efficacy Safety and Tolerability of Azelaprag (BGE-105) to Prevent Acute Skeletal Muscle Atrophy in Participants Hospitalised with an Acute Exacerbation of Chronic Obstructive Pulmonary Disease (AECOPD) |
BioAge Labs Inc. | Recruitment not yet started |
| 1009199 |
A Phase I/IIa Open-label Modular Dose Escalation and Dose Expansion Study to Evaluate the Safety Pharmacokinetics Pharmacodynamics and Efficacy of AZD2796 an Anti-LILRB2 Antibody as monotherapy (or in combination) in Adult Patients with Advanced Solid Tumors |
Astra Zeneca AB | Recruitment not yet started |
| 1009269 |
A Phase 2/3 Randomized Double-blind Controlled Study of Zanzalintinib (XL092) in Combination with Pembrolizumab vs Pembrolizumab in the First-line Treatment of Subjects with PD-L1 Positive Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma |
Exelixis Inc. | |
| 1009289 |
A Phase 2a Open-Label Multiple Dose Study Evaluating the Safety Tolerability and Pharmacodynamics of Imdusiran (AB-729) in Combination with Intermittent Dosing of Durvalumab a PD-L1 Monoclonal Antibody in Subjects with Chronic HBV Infection |
Arbutus Biopharma Corporation | Recruitment not yet started |
| 1009423 |
A Phase 3 Randomized Double-Blind Placebo-Controlled 3-Arm 3-Period Study to Assess the Efficacy and Safety of a New Formulation of Oral Cladribine Compared with Placebo in Participants with Generalized Myasthenia Gravis |
Merck Sharp & Dohme LLC | |
| 1009489 |
A Phase 1b Study to Assess the Safety, Tolerability, and Pharmacokinetics of ION269 in Adults with Down Syndrome and Adults with Early Symptomatic Alzheimer’s Disease (HERO Study) |
Ionis Pharmaceuticals Inc. | Recruitment not yet started |
| 1009575 |
A Phase 2 randomized placebo-controlled double-masked dose-finding study to investigate the efficacy and safety of BAY3283142 in patients with moderately severe to severe non-proliferative diabetic retinopathy (ARGON-NPDR) |
Bayer AG | Recruitment not yet started |
| 1009611 |
A Phase III, Randomized, Open-Label, Multicenter Study Evaluating the Efficacy and Safety of Divarasib Versus Sotorasib or Adagrasib in Patients With Previously Treated KRAS G12C-Positive Advanced or Metastatic Non-Small Cell Lung Cancer |
F. Hoffmann- La Roche AG | |
| 1009644 |
A phase 1/2 study of Linvoseltamab in patients with relapsed or refractory systemic light chain amyloidosis |
Regeneron Pharmaceuticals Inc. | |
| 1009713 |
A Randomised, Double- Blind, Placebo-Controlled, Phase 2 Study to Assess the Prophylatic AntiViral Activity Human RhinoVirus, Safety, And Tolerability of HEX17 (NEUMIFIL) in Healthy Adult Participants Via A Human Viral Challenge Model |
Pneumagen Limited | |
| 1009715 |
A Multicenter Open-label Phase 1/2 Dose-escalation and Subsequent Safety Extension Study of Subcutaneous KK8123 in Adult Patients with X-linked Hypophosphatemia |
Kyowa Kirin Inc. | |
| 1009774 |
A Randomized Single-Blind Placebo-Controlled Oral Multiple Ascending Dose Study to Evaluate the Safety Tolerability and Pharmacokinetics of MM402 in Healthy Volunteers |
Mind Medicine Inc | Recruitment not yet started |
| 1009776 |
Phase 3 Double-blind Multicenter Randomized Active-Controlled Study to Evaluate the Safety and Efficacy of Bictegravir/Lenacapavir Versus Biktarvy ® (Bictegravir/Emtricitabine/Tenofovir Alafenamide) in Virologically Suppressed People With HIV-1 |
Gilead Sciences Inc. | |
| 1009816 |
An Open-Label Extension Study to Evaluate the Long-Term Safety and Efficacy of Once Daily Mexiletine PR in Patients with Myotonic Dystrophy Type 1 and Type 2 who have completed the MEX-DM-302 Study |
Lupin Atlantis Holdings SA | |
| 1009838 |
A Phase 3 Study of Pembrolizumab in Combination With Carboplatin/Taxane (Paclitaxel or Nab-paclitaxel) followed by Pembrolizumab With or Without Maintenance MK-2870 in the First-line Treatment of Metastatic Squamous Non-small Cell Lung Cancer |
Merck Sharp & Dohme LLC | |
| 1010057 |
A randomized multicenter double-blind Phase 3 study to investigate the safety and efficacy of belrestotug in combination with dostarlimab compared with placebo in combination with pembrolizumab in participants with previously untreated unresectable locally advanced or metastatic PD-L1 selected non small cell lung cancer (GALAXIES LUNG 301) |
GlaxoSmithKline Research & Development Limited |
Recruitment not yet started |
| 1010098 |
A phase 1 randomized double-blind placebo controlled parallel group study of the safety tolerability and pharmacokinetics of single doses of GSK3923868 administered via dry powder inhaler to healthy participants of Chinese Japanese and European ancestry |
GlaxoSmithKline Research & Development Limited |
|
| 1010123 |
The ENERGY 2 Study: An Open-Label Phase 3 Study to Evaluate the Efficacy and Safety of INZ-701 in Infants with Ectonucleotide Pyrophosphatase/Phosphodiesterase 1 (ENPP1) Deficiency |
Inozyme Pharma Inc. | Registration currently underway |
| 1010222 |
A Multicenter Randomized Double-Blind Placebo-controlled Study to Assess the Efficacy and Safety of Treatment with Bepirovirsen in Participants living with Human Immunodeficiency Virus and Chronic Hepatitis B Virus Infection on Antiretroviral Treatment |
GlaxoSmithKline Research & Development Limited |
|
| 1010309 |
A Phase Ib Multicenter Non-Randomized Open-Label Study to Investigate the Safety Tolerability Pharmacokinetics Immunogenicity and Pharmacodynamics of Single Ascending Doses of a New Subcutaneous Formulation Of Ocrelizumab in Patients With Multiple Sclerosis |
F. Hoffmann- La Roche AG | Registration currently underway |
| 1010357 |
An open-label multi-center phase I/II study to assess safety efficacy and cellular kinetics of YTB323 in participants with treatment-resistant generalized myasthenia gravis |
Novartis Pharma AG | |
| 1010387 |
A Two-Part Study to Assess the Safety Tolerability Pharmacokinetics and Pharmacodynamics of Single and Multiple Ascending Oral Doses of PTD802 and to Characterise the Effect of Food on the Pharmacokinetics of PTD802 in Healthy Male and Female Subjects |
Pheno Therapeutics Limited | Recruitment not yet started |
| 1010431 |
A Phase 3 Global Randomized Modified Double-Blind Placebo-Controlled Study to Evaluate the Efficacy Immunogenicity and Safety of IVX-A12 a Respiratory Syncytial Virus (RSV) and Human Metapneumovirus (hMPV) Virus-Like Particle (VLP) Vaccine in Adults 60 Years of Age and Older |
Astra Zeneca AB | Recruitment not yet started |
| 1010538 |
Long-term efficacy and safety of cagrilintide s.c. 2.4 mg in combination with semaglutide s.c. 2.4 mg (CagriSema 2.4 mg/2.4 mg) once weekly versus placebo in participants with obesity |
Novo Nordisk A/S | |
| 1010692 |
A Phase 2a Open-label Multicenter Platform Trial to Assess the Safety Tolerability and Efficacy of Inebilizumab and Blinatumomab in Subjects With Select Autoimmune Diseases (Master Protocol) |
Amgen Inc. |
The information in the above table is also available in a CSV file.
Research transparency requirements under the new clinical trials regulations
At the moment, it is a condition of a Research Ethics Committee (REC) favourable opinion that a clinical trial is registered on a publicly accessible registry.
From 28 April 2026, the amended UK clinical trials regulations will come into effect, which will feature specific requirements for research transparency.
- register clinical trials in a public registry before the recruitment of the first participant or within 90 days of approval of the clinical trial (whichever is sooner)
- publish a summary of trial results within 12 months of completion
- offer to share a summary of results with participants in a format they can easily understand
Read more about the specific requirements relating to research transparency
Research transparency data reporting
We have also published data on whether trials that completed in 2023 published results on a public registry and shared a summary of results with their participants.