Transforming the UK's clinical trial landscape: updated regulations are now in force
The Health Research Authority (HRA) and the Medicines and Healthcare products Regulatory Agency (MHRA) have introduced the largest package of clinical trial regulatory reforms in over 20 years.
The updated regulations are designed to protect trial participants, strengthen patient safety, and accelerate approvals by reducing unnecessary burdens on researchers, to support high-quality, trusted research taking place in the UK.
Feedback from a public consultation in 2022 helped shape the proposals to improve how clinical trials of investigational medicinal products (CTIMPs) were run in the UK.
Working in partnership, the HRA and MHRA sought the views of patients, researchers, healthcare professionals and industry to inform and develop the new framework.
The Medicines for Human Use (Clinical Trials) (Amendment) Regulations 2025 were signed into law in April 2025.
Following a 12-month implementation period, the regulations came into force on 28 April 2026.
Who these changes affect
The updated regulations impact everyone involved in CTIMPs in the UK:
- researchers and sponsors
- Research Ethics Committees (RECs)
- trial participants and the public
- healthcare professionals
Why these changes matter
The reforms:
- align the process for the MHRA and RECs to review and approve applications and modifications
- enhance transparency and accountability for research findings
- reduce unnecessary burdens while upholding ethical and safety standards
Together, these improvements are designed to strengthen the UK's position as a global leader in clinical research.
Key changes in the regulations
The following areas of the regulations have been updated.
New and updated definitions
The term ‘amendment’ to describe changes to approved trials has been replaced with ‘modification’. This term is already used by European Union nations to describe changes to CTIMPs. This change makes sure there is better alignment between the UK and the international research community.
Modifications are now categorised as ‘substantial modifications’, ‘modification of an important detail’ or ‘minor modifications’.
The updated regulations have introduced new definitions including ‘non-investigational medicinal product’, ‘notifiable trial’ and ‘public registry’.
There have also been changes to existing definitions including the removal of the term ‘authorised health care professional’. Instead, the updated regulations say that chief investigators and investigators should be a health care professional as defined in the amended legislation.
The updated regulations also replace the term ‘subject’ with ‘participant’ and ‘trial site’ with ‘trial location’.
Read more about the new and updated terms in our blog.
The approvals process for clinical trials
Combined Review – the system that lets researchers apply for ethics and regulatory approval in one go – and a new streamlined notification scheme for some clinical trial initial applications and modifications, have been written into law as part of the updates.
The updated regulations have changed how Research Ethics Committees (RECs) and the MHRA process applications and modifications for CTIMPs, including for Phase 1 healthy volunteer trials.
Under the amended regulations, the MHRA and RECs can request that sponsors modify their trials in certain circumstances.
The legislation also sets the expectation that all CTIMPs should recruit their first participant in the UK within 2 years of the trials being approved, with extensions available. If there is no recruitment within 2 years and an extension has not been granted, the approval will lapse.
Transparency
For the first time in the UK, it is a legal requirement to:
- register clinical trials involving medicines in a public registry
- publish a summary of trial results within 12 months of completion
- offer to share a summary of results with participants (or other relevant people) in a way they can easily understand
Provisions for deferrals in specific circumstances, such as phase 1 trials involving healthy volunteers, are available.
Research Ethics Committees (RECs)
Changes to RECs are designed to improve flexibility while aligning with international good clinical practice (ICH-GCP E6). Each REC must meet the following requirements:
- need at least 5 members that collectively possess the qualifications and experience to review and evaluate any proposed trial's scientific, medical, and ethical aspects
- still be expected to have an appointed Chair
- include 1 lay member
Simplified arrangements for consent in clinical trials
The updated clinical trials regulations offer sponsors of clinical trials that meet certain conditions the option to use simplified arrangements for seeking and evidencing informed consent.
We’ve been working with an advisory group to help us develop principles that will support sponsors and research team members to use simplified arrangements when seeking and evidencing informed consent in low intervention clinical trials.
These principles are intended to support proportionate approaches to the information provided to participants, the conduct of consent discussions, and the methods used to evidence consent, ensuring that consent remains informed, freely given, and explicit.
The principles have now been published and are available as part of our suite of clinical trials regulations guidance.
Guidance instead of legislation
Some areas are being supported through detailed guidance rather than new legal requirements.
We are leading work on guidance to support best practices in diversity and inclusion, and public involvement in clinical trials.
Diversity and inclusion in clinical trials
Including a wider range of participants ensures trial findings reflect the entire population's needs.
Our Public Perceptions of Research report found that:
- 88% believe trials should involve a diverse group of participants.
- 70% support this even if it increases costs
- 74% are in favour even if it extends timelines
We've developed a draft set of questions and supporting guidance jointly with the Medicines and Healthcare products Regulatory Agency (MHRA) for researchers to consider when they design clinical trials and clinical investigations. The answers to these questions will form the basis of an Inclusion and Diversity Plan.
This will help make sure clinical research is designed to include people who could be impacted by the findings, and that people underserved by research are not overlooked.
Following an informal consultation on the draft questions and supporting guidance in 2024, we ran a pilot in which sponsors and researchers were invited to submit a plan as part of their IRAS application.
The pilot closed in December 2025. We’re gathering and reviewing feedback from the sponsors, researchers and ethics committee members who took part, and plan to publish the findings and set out our next steps soon.
The new guidance we’re developing for diversity and inclusion is being informed by the clinical trials regulations, however it is not a legal requirement and is not part of the suite of clinical trials regulations guidance which must be followed from 28 April 2026.
Public involvement
Involving the public in designing, delivering, and reporting clinical trials leads to better outcomes and builds trust.
We have updated our website to:
- provide clear expectations for public involvement in health and social care research, including all phases of clinical trials
- point to the wide range of resources available across the sector
The updates are designed to help researchers meaningfully involve the public in all clinical trials.
Timeline of key changes
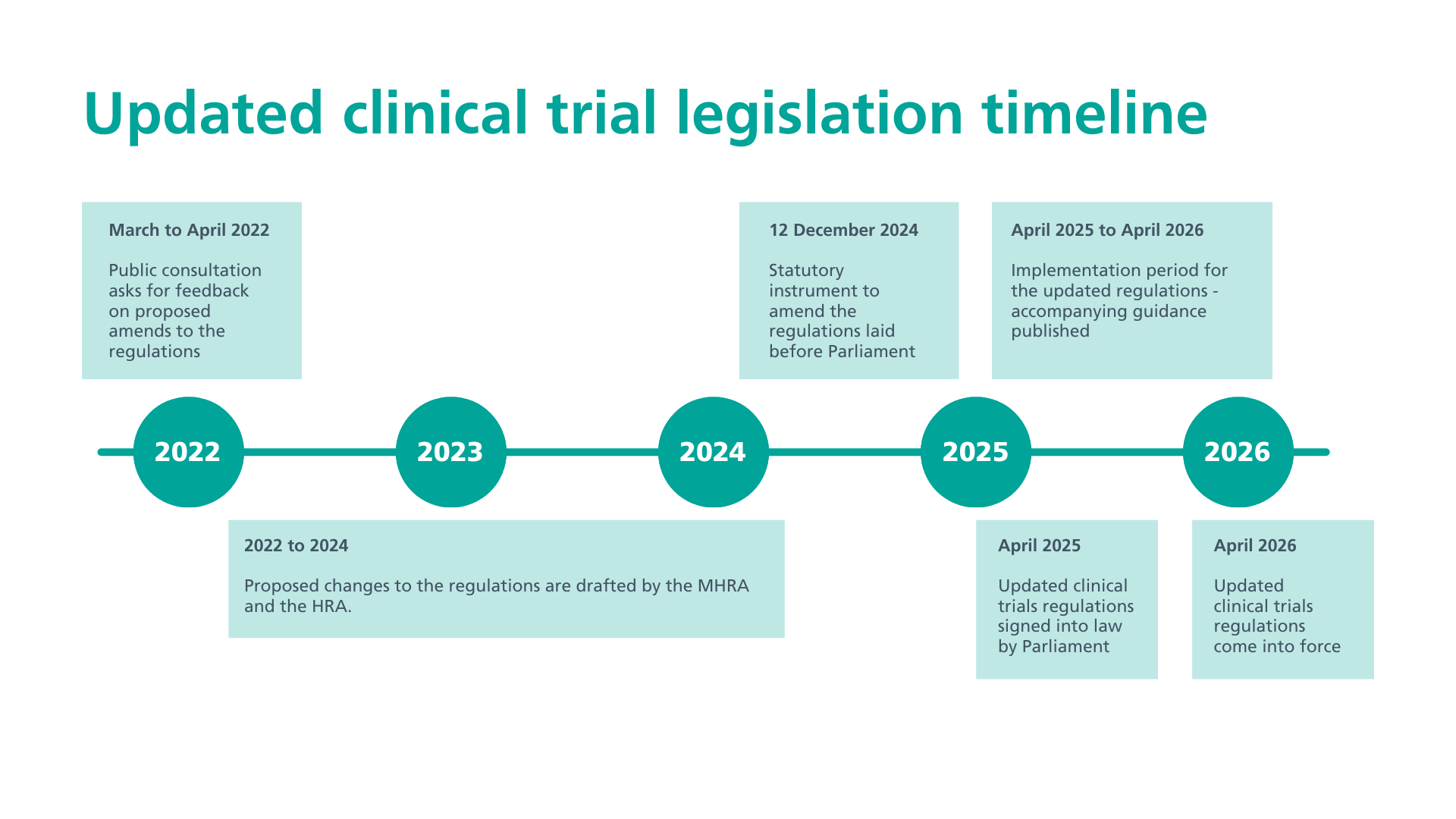
Milestones
- March to April 2022 - public consultation
- December 2024 - Statutory Instrument laid before Parliament
- February 2025 - approved by Westminster Parliament and House of Lords
- April 2025 - approval from the Northern Ireland Assembly and final ministerial sign off
- June 2025 - new guidance is published to accompany the new regulations
- 1 October 2025 - final guidance to accompany the regulations is published
- 28 October 2025 - 6 months until clinical trials regulations come into force
- 28 April 2026 - updated clinical trials regulations come into force
Support for researchers and sponsors
We've worked closely with our stakeholders during the implementation period to produce a range of support:
- guidance – see guidance section below
- training – see webinar section below
- newsletter – our Clinical Trials update gives you the latest updates and information on the updated clinical trials regulations
- news and blogs – sharing the latest updates on our website
Guidance to accompany clinical trials regulations
We’ve published guidance for researchers and sponsors explaining the changes to the regulations.
Our guidance covers the following updates which have been a key focus for the HRA:
- definitions and terminology
- pharmacovigilance
- the approvals process for clinical trials
- Research Ethics Committees that review clinical trials
- simplified arrangements for consent in clinical trials
- research transparency requirements for clinical trials
The guidance outlines the review and assessment processes for CTIMPs by RECs and the MHRA.
MHRA guidance
The MHRA has also published separate guidance to accompany the amended regulations which you can read on their clinical trials hub.
Changes to non-CTIMP studies
We've introduced changes to how other types of health and social care research (non-CTIMPs) are processed and managed to align with the updated clinical trials regulations.
This is designed to provide consistency across all UK clinical research and to streamline the regulatory process, making it simpler for sponsors and researchers to earn approvals.
The HRA has published guidance on the changes to non-CTIMP clinical trials and other types of research.
Webinars
The HRA and MHRA held a Clinical Trials Regulations: Countdown to Implementation webinar webinar on 12 March.
The webinar covered:
- the key changes in the updated regulations
- transitional arrangements
- HRA and MHRA guidance
- question and answer session
Useful links
- MHRA – clinical trials regulation
- NIHR – supporting research in the UK
- EU clinical trials regulation (CTR)
Sign up for updates on the clinical trial regulations

We send out regular email updates about the amended clinical trials regulations in the UK.
These emails include updates on the different areas the HRA is leading on, and support for researchers and sponsors.
You can take a look back at previous editions on our clinical trials update webpage.
Clinical trials regulations news
Visit our clinical trials regulations news and updates page for a round up of our work on the regulations.