Our vision for research transparency is that trusted information from health and social care research studies is publicly available for the benefit of all. Publishing results and sharing a summary of results with participants are crucial to ethical research practice and avoiding research waste.
The UK Policy Framework for Health and Social Care Research says:
‘Other than research for educational purposes and early phase trials, the findings, whether positive or negative, [should be] made accessible, with adequate consent and privacy safeguards, in a timely manner after they have finished.’
It also says that ‘Information about the findings of the research [should be] available, in a suitable format and timely manner, to those who took part in it, unless otherwise justified.’
We currently ask researchers to notify us that study results have been published and that results have been fed back to the participants via the final report.
All project-based research (not research tissue banks or research databases) that has been reviewed by a Research Ethics Committee (REC) needs to submit a final report. This is listed in the Standard Conditions issued with decision letters issued to sponsors.
This final report should be submitted to the REC within a year of the global end of trial date and helps the HRA determine whether the research was conducted in accordance with the REC favourable opinion. The summary of results provided is also used to update our research summaries pages.
What we are publishing
We are sharing the percentages of sponsors that have published, or told us they have published, their study results on a public registry, as well as the percentages that have shared or are in the process of sharing their results with participants, and if they have told us they have done so.
The data covers clinical trials in the UK that completed in 2023, including whether they have submitted a final report within a year which is our way of measuring compliance.
From next year, we will be sharing more details on the compliance of sponsors with our transparency expectations, such as a list of those who have told us they have not published their summary of results on a public registry, or have not told us whether they have published because they have not submitted a final report. Next year we will share data on trials that were completed in 2024.
Clinical trials are defined as:
- clinical trial of an investigational medicinal product
- clinical investigation or other study of a medical device
- combined trial of an investigational medicinal product and an investigational medical device
- other clinical trial to study a novel intervention or randomised clinical trial to compare interventions in clinical practice
Please note that in our data published values have been rounded and may not add up to 100%.
What we found
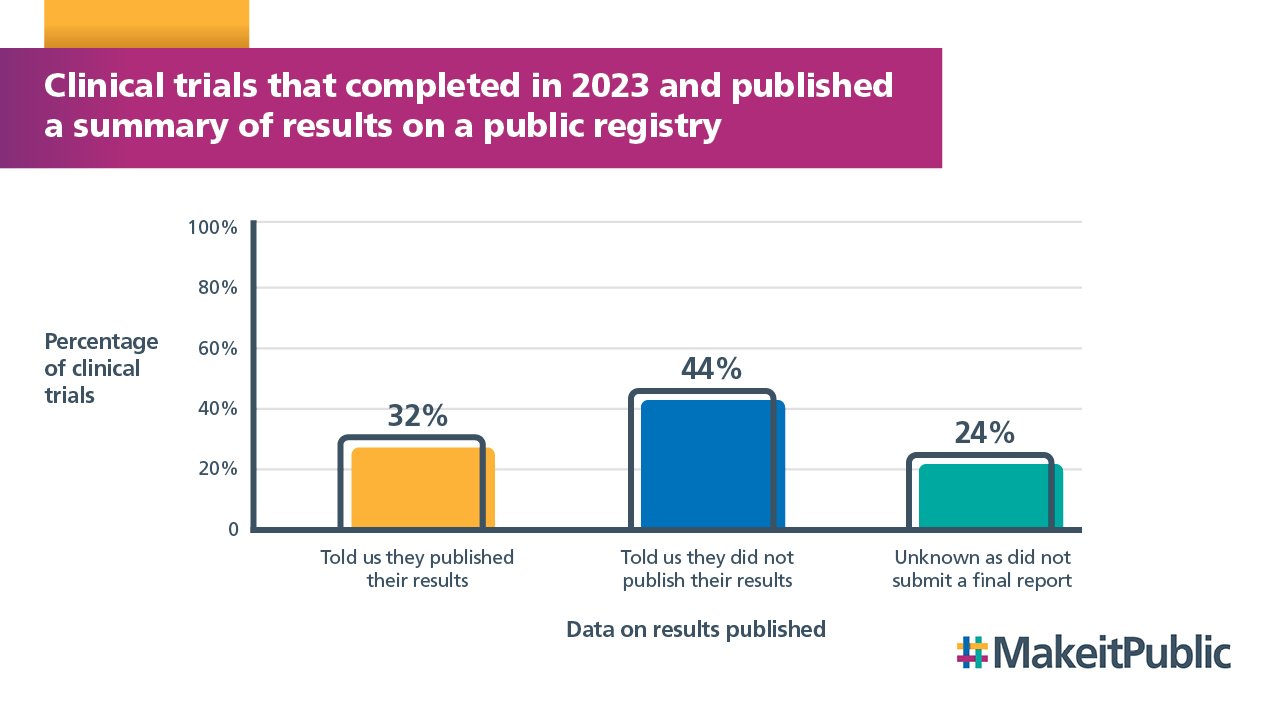
32% of clinical trials told us they had published results on a public registry, whilst 44% told us they had not published their results.
We did not receive a final report for 24% of trials completed in 2023 so we do not have this data.
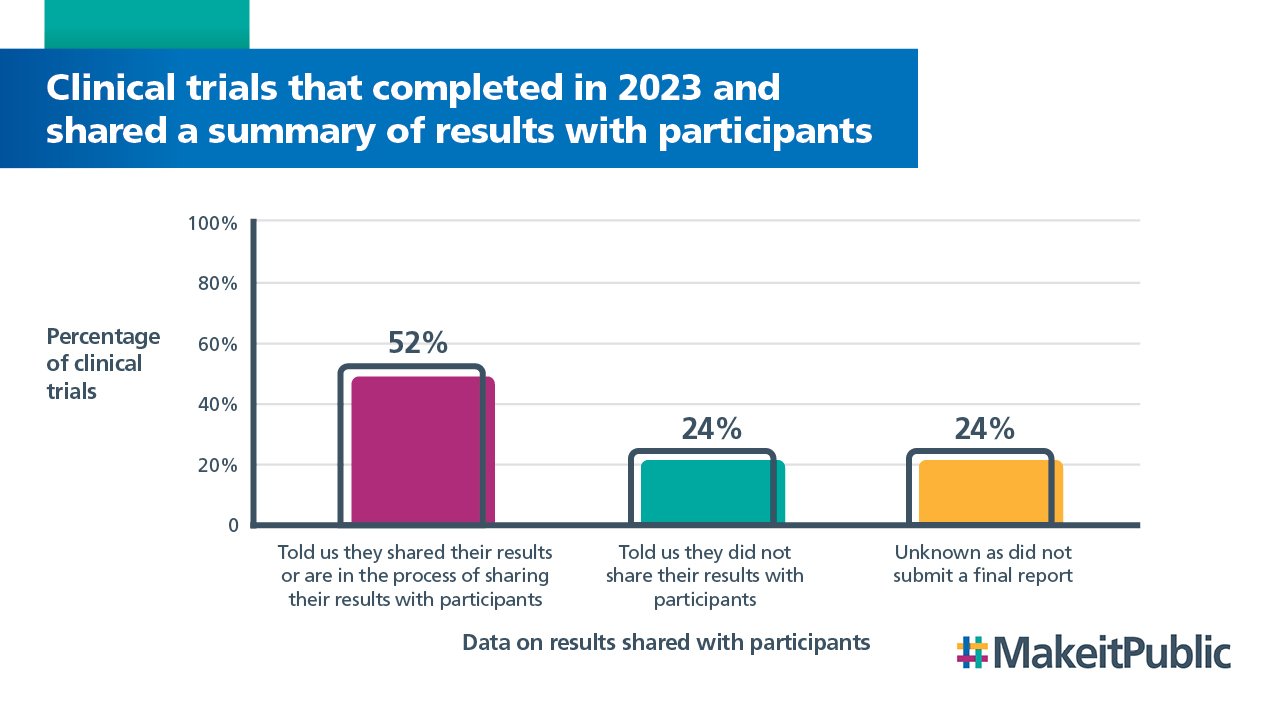
52% of clinical trials told us that they had shared, or are in the process of sharing a summary of their results with participants.
24% told us that they did not share their results with participants.
We did not receive a final report for 24% of trials completed in 2023 so we do not have this data.
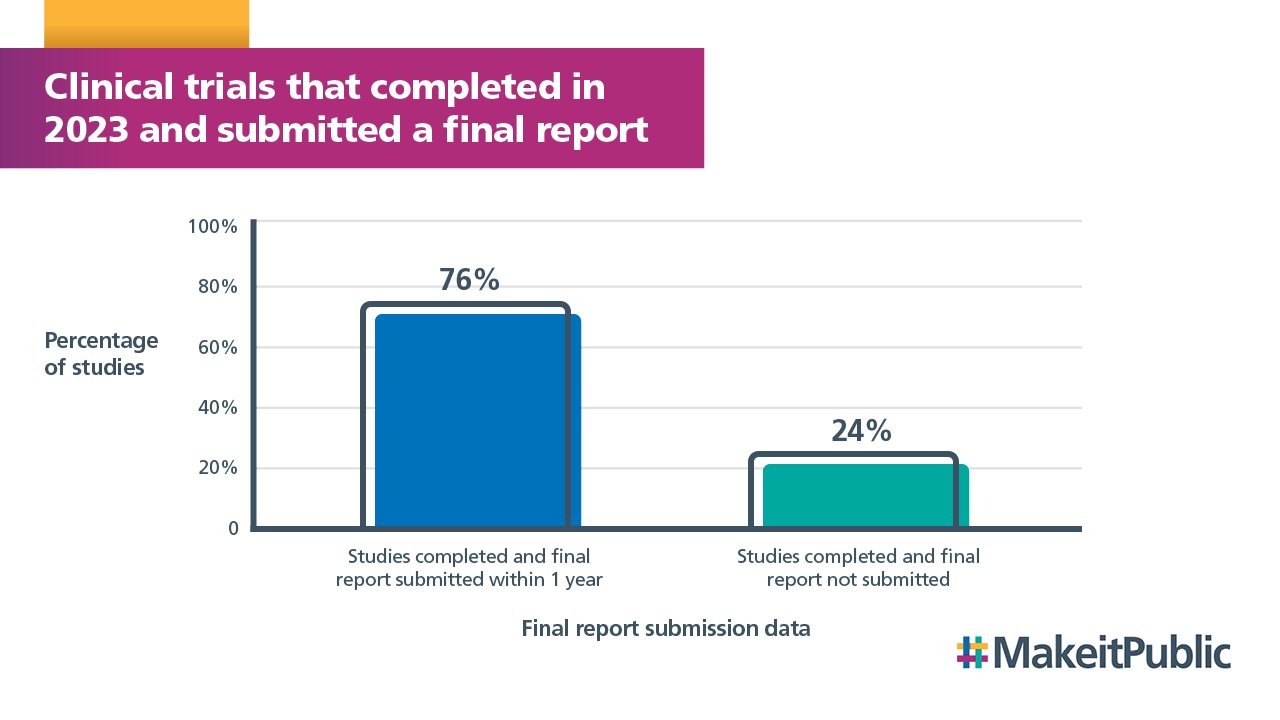
At the time of publication of this report, 76% of clinical trials that were reported to us as completed in 2023 provided the REC with a final report.
Work was undertaken with sponsors who had not submitted their final report which increased the response rate by 8% from 68%. However, 24% of studies did not submit their final report within the required timeframe.
Research transparency requirements under the new clinical trials regulations
From 28 April 2026, the amended UK clinical trials regulations will come into effect, which will include legislation on research transparency.
It will become a legal requirement to:
- register clinical trials in a public registry before the recruitment of the first participant or within 90 days of approval of the clinical trial (whichever is sooner)
- publish a summary of trial results within 12 months of completion
- offer to share a summary of results with participants in a format they can easily understand
Find out more about the clinical trials regulations.
We’re also making changes to non-CTIMP clinical trials and other types of research to align with the regulations and to streamline the applications process.
Research transparency data reporting
We have also published data on the registration status of clinical trials that received a REC favourable opinion in 2024. Read the report.