Combined Review is the way research teams seek approval for new clinical trials of investigational medicinal products (CTIMPs) and combined medicine and device trials.
Research teams make a single application using a new part of IRAS, which goes to both the Medicines and Healthcare products Regulatory Agency (MHRA) and a Research Ethics Committee (REC) at the same time. The application also goes for study wide review, such as HRA and HCRW approval, if the study is to take place in the NHS or Northern Ireland HSC.
The regulatory and ethics reviews are done in parallel and any requests for further information (RFIs) are raised jointly. A single response to these requests leads to a single decision from both reviews. Study wide review is usually issued at the same time as MHRA and REC, but may come later if there are still issues to discuss with the applicant.
IRAS is used for the initial application and supports the trial through modifications right up to the end of the trial. Research teams can allocate different roles in the system for colleagues working on the project.
In exceptional circumstances, submitting separate applications to the MHRA and a REC may be acceptable. This is only permitted where it would otherwise not be possible to submit a trial application in the UK.
If a sponsor believes they have an application that needs separate applications, they should contact the MHRA at clintrialhelpline@mhra.gov.uk. In the email, they should give details about the trial and explain why they believe separate applications are needed. Requests are assessed on a case-by-case basis, and if agreed, instructions will be given on how to submit the application.
Initial application timeframes and process
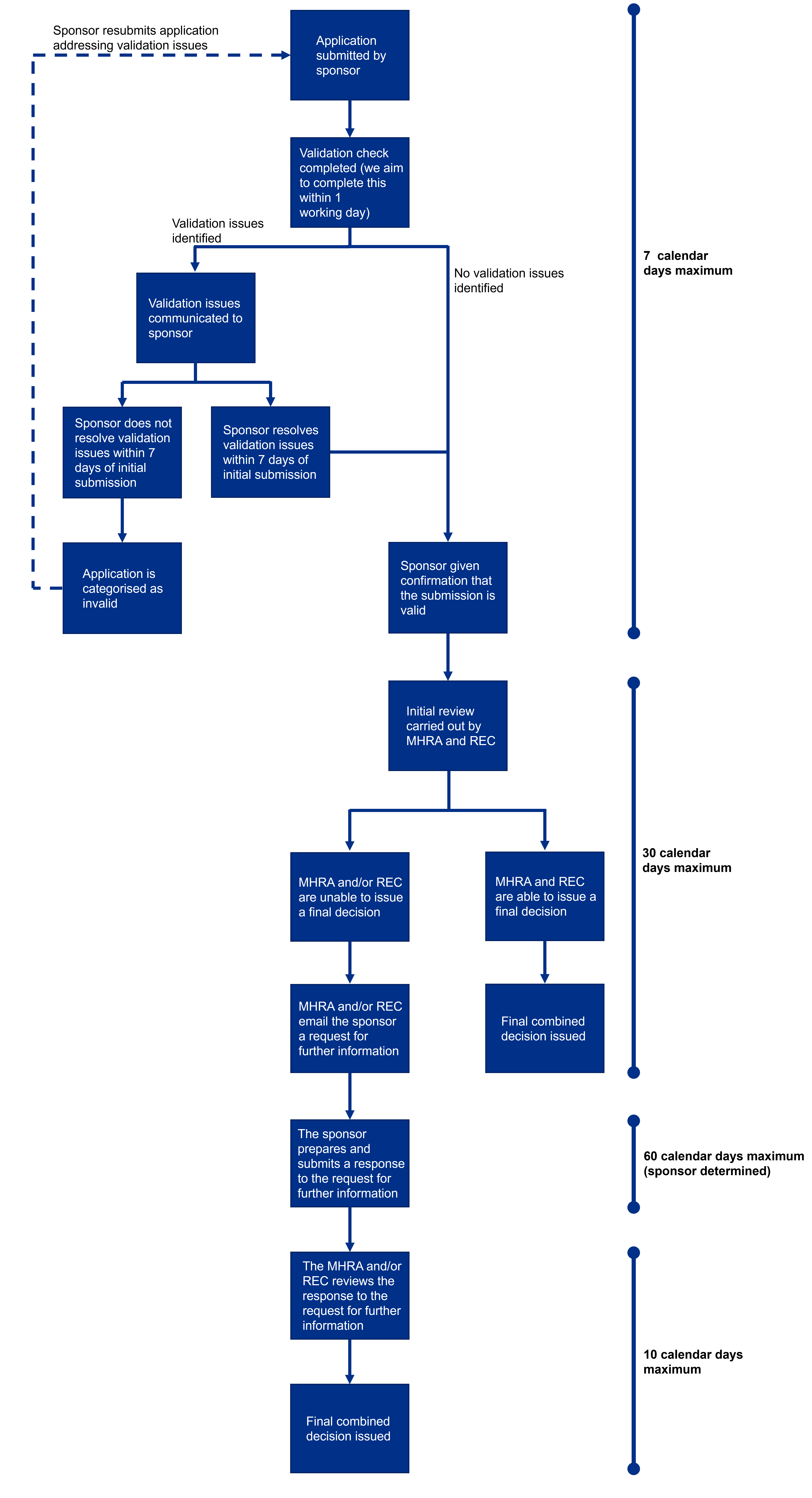
The REC outcome will be issued as soon as possible after the REC meeting. This is so that any changes which are required to supporting documentation or any points of further clarification can start to be worked on. If the REC raises any issues which relate to the scope of the MHRA review, these issues are provided to the MHRA for consolidation; and will therefore be issued when the MHRA outcome is issued.
The sponsor should wait for the full request for further information to be issued in the Integrated Research Application System (IRAS) before responding to any points they receive individually from the MHRA or REC. This is because the full request for further information will have the final consolidated feedback, including any queries or issues, from both the MHRA and REC.
The MHRA or REC will issue a final combined decision or, if they cannot reach a final decision, they will request further information from the sponsor within a maximum of 30 calendar days from the application being confirmed as valid.
If the MHRA or REC send a request for further information, the sponsor will have up to 60 calendar days to provide a response. The sponsor can respond at any point within this timeframe.
If sponsors need longer than 60 days to respond, they can ask for an extension. Sponsors can do this by contacting the MHRA at clintrialhelpline@mhra.gov.uk or, if the points raised only relate to the REC review, by contacting the REC directly. In their request, the sponsor should explain why they need an extension and when they expect to respond. If an extension is agreed, the REC and MHRA will make each other aware of this.
After a response to a request for further information is received, the REC and/or MHRA will review the response and give the final outcome.
This will be done within a maximum of 10 calendar days of the MHRA or REC receiving the response.
Independent expert advice
For some applications, the REC or MHRA may need independent expert advice from a specialist group or committee.
For information on when independent expert advice may be needed, read the MHRA's guidance. Their guidance also explains what a sponsor should do if they suspect their application may need independent expert advice.
When independent expert advice is needed, it will be sought during the initial review of an application and/or after receiving a response to a request for further information. The timeframes for the approval process may be extended to allow for the advice to be requested and received.
If independent expert advice is sought:
- during the initial review stage for your application, the 30 day timeframe may be extended by up to an additional 90 calendar days (bringing the maximum timeframe to 120 calendar days)
- following a response to a request for further information being received, the 10 day timeframe may be extended by up to an additional 30 calendar days (bringing the maximum timeframe to 40 calendar days)
- following a response to a request for further information being received and an advanced therapy medicinal product (ATMP) is involved in the trial, the 10 day timeframe may be extended by up to an additional 60 calendar days (bringing the maximum time frame to 70 calendar days)
In all cases, we’ll make sure it proceeds as quickly as possible to avoid unnecessary delays in sponsors receiving the outcome.
If the REC or MHRA determine that they need to seek independent expert advice, they'll notify the sponsor, ask the sponsor for any additional information needed, and let the sponsor know when they can expect to receive the outcome.
Timeline for xenogenic cell therapy applications
If a sponsor submits an application involving xenogenic cell therapy, the same review process is followed. Both the MHRA and REC will review the application as efficiently as possible, as they would any other application. However, the above timeframes will not apply. This means that outcomes and decisions can be issued at any point after an application is received and validated.
Quick Start Guide
Getting started
- first-time user? Contact us at cwow@hra.nhs.uk for help to get started.
- check if you need to create an account
- sponsor and Sponsor Delegate Organisations are selected from options within IRAS - if yours is not listed, contact our Service Desk
- access early study support and advice services
- get help with new and ongoing applications with our new IRAS user guide or the web step by step guide
- read more about the benefits
Further guidance and support
- Pharmacy and Radiation Technical Assurance Submission Guidelines
- instructions for studies with ionising radiation
- how and when to submit to ARSAC
- guidance for combined IMP and device trials
- information on registration of studies
- current legislation and regulations
- REC directory to search meeting dates (filter by RECs recognised to review CTIMPs)