Being transparent about your research builds trust.
Making information about your research publicly available is vital to ethical research practice.
When research is carried out openly and transparently, everyone will be able to see what research is happening and the outcomes from finished studies.
This means that:
- patients, service users and carers can find opportunities to join studies
- researchers can build on previous research findings in their efforts to develop new and better treatments, and to identify the best ways for us to stay healthy and well
- health professionals, commissioners, policy makers and funders can use research findings to help make informed decisions
If you work in research, it is important that you are aware of what you are legally required or expected to do when it comes to transparency.
What to do if you work in research
The UK Policy Framework for Health and Social Care Research sets out the responsibilities for researchers, sponsors and funders for all aspects of research.
The framework outlines four key areas of responsibility when it comes to research transparency.
These are:

Planning your research
Research transparency should be considered at each step of the research process, from the design and planning stage, right through to completion. The flowchart below shows each stage and the boxes in pink are the stages at which you can promote research transparency.
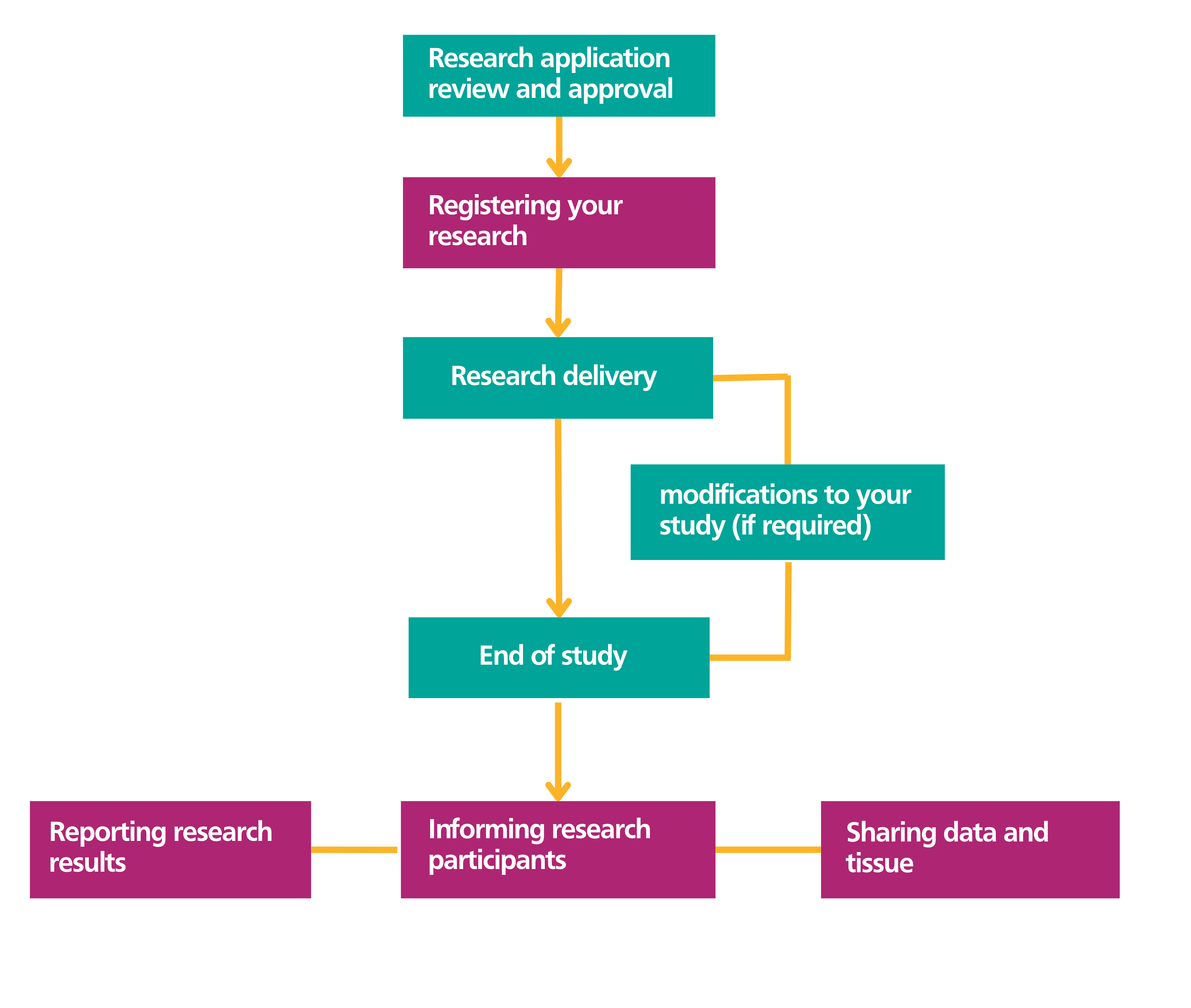
Guidance for those working in research
We've developed guidance to make it easy to understand what you need to do to be transparent in each of the 4 key areas highlighted in pink in the above flow chart.
The HRA's role in research transparency
The Health Research Authority has a legal duty to promote research transparency in the UK.
Find out what we do to make transparency the norm in research.
Make it Public
Make it Public is a campaign dedicated to research transparency.
The Make it Public group brings together people and organisations across the research community to champion research transparency across the sector.
The group meets quarterly to discuss how to improve research transparency and highlight good practice.