Registering your research in a public registry is the first thing you should do when it comes to transparency.
Amended clinical trials regulations, which came into force on 28 April 2026, introduce new legal requirements for research transparency in clinical trials of investigational medicinal products (CTIMPs) in the UK.
They require sponsors of CTIMPs to:
- register a clinical trial in a public registry either before the date on which the first participant is recruited (when they sign the consent form) or within 90 calendar days of approval of the clinical trial – whichever comes first
For clinical trials which are not regulated under the clinical trial regulations, it is a condition of a favourable ethics opinion.
It is good practice for all other studies.
Registering trials reduces research waste. It prevents duplication and means that more people can find out about studies to join.
Trusted information about health and social care studies should be publicly available for the benefit of all, and registration is the first step.
Information and guidance on registering your research
Guidance on each area of research registration is included below. Click on each topic to jump to the information and guidance:
Where you need to register your clinical trial
When to register your clinical trial
What details you should include when registering your clinical trial
What happens if you abandon the clinical trial
The process for registering a trial submitted through Combined Review
What to do if you have already registered or plan to register your trial on ClinicalTrials.gov
Registration of research tissue banks
Information about your clinical trial that is shared on the HRA website
Deferral of registration on a public registry
Deferral of research summary on the HRA website
Where you need to register your clinical trial
You need to register your clinical trial in a public registry.
This means any registry on the World Health Organisation (WHO) list of primary registries that facilitates public access to information about the UK trial.
We ask sponsors to use either ISRCTN or ClinicalTrials.gov as these both feed in to Be Part of Research. This is a free service which makes it easy to find and take part in vital health and care research across the UK.
This also helps bring us closer to having a full record of all UK clinical trials in a single place.
Please note that for trials taking place in both the EU and the UK, registration with the EU Clinical Trials Information System (CTIS) will not satisfy the requirement for registration as it does not support visibility of UK trials.
How to know if your research is a clinical trial that is regulated by the clinical trials regulations
This is research investigating the safety and efficacy of a medicinal product:
- a clinical trial of an investigational medicinal product (CTIMP)
- a combined trial of an investigational medicinal product and an investigational medical device (see information on Combined Review)
How to know if your research is a clinical trial that is not regulated by the clinical trials regulations
This is a clinical trial not investigating the safety and efficacy of a medicinal product:
- a clinical investigation or other study of a medical device
It can also be any other clinical trial to study a novel intervention or randomised clinical trial to compare interventions in clinical practice, for example research involving:
- trials of surgical interventions
- radiotherapy
- imaging investigations
- mental health investigations or therapies
- physiological investigations
- trials of products not defined as medicines or medical devices (for example nutritional)
- complementary or alternative therapies
When to register your clinical trial
You should register your trial either before the date on which the first participant is recruited (when they sign the consent form) or within 90 calendar days of approval of the clinical trial – whichever comes first.
What details you should include when registering your clinical trial
If your trial is already registered when you complete your IRAS application, you should include your registration number.
If you register after submitting your IRAS application, you should email the Research Ethics Committee and deferrals@hra.nhs.uk with your registration number as soon as possible.
You should ensure you keep the record in the registry up to date.
If you make modifications to your study, you should update the public registry as needed.
What happens if you abandon the clinical trial
If you decide to abandon a clinical trial before it commences (before any trial procedures such as patient screening or consent set out in the protocol have been initiated), then the trial will not need to be registered.
The process for registering a trial submitted through Combined Review
If you submitted your research application through Combined Review, then your study information will be sent directly to ISRCTN for registration on your behalf. This is due to a partnership we have with them to make the process easier for you.
This is the case for all clinical trials of investigational medicinal products (CTIMPs). ISRCTN will be in touch with you to ask for some additional information to finalise the registration. ISRCTN will also let us know the registration number for your trial, so you don’t need to email us.
You should still make sure that the record of your trial in the registry is kept up to date as the study progresses.
Find out more about our partnership with ISRCTN.
Find out more about the information we share with ISRCTN registry.
What to do if you have already registered or plan to register your trial on ClinicalTrials.gov
If you tell us that you have registered with ClinicalTrials.gov or that you would prefer to register your clinical trials here, then you can request that your study is not registered with ISRCTN.
To let us know that you have or will register on ClinicalTrials.gov, you’ll need to make this clear in your IRAS application. You should do this in ‘section c’ of the study information section of your Combined Review application in IRAS.
If you have not included your ClinicalTrials.gov registration number in your IRAS application, you should email it to the REC and deferrals@hra.nhs.uk as soon as possible.
Registration of research tissue banks
It is a condition of a favourable ethics opinion that all research tissue banks are registered on the UK Clinical Research Collaboration (UKCRC) Tissue Directory.
The research tissue bank should be registered no later than 90 calendar days after favourable opinion, or 90 calendar days after the research tissue bank holds tissue with the intention to provide for research purposes.
Registration means having added details of the types of tissue samples held in the tissue bank. Your research registration reference number should be entered in your dataset on the Integrated Research Application System (IRAS), so that this can be included on your application form.
See the Research Tissue Bank webpage for further information.
Information about your clinical trial that is shared on the HRA website
We publish details of all research in the UK that is reviewed by a REC or seeking HRA approval in the research summaries section of our website.
This includes information about the planned research, a summary of the research as submitted to the REC, and the REC’s opinion.
We also publish details of all applications approved by the Confidentiality Advisory Group (CAG) in the CAG registry. This contains summary information about the activity, details of the identifiers approved and applicant contact details.
This information is pulled through from your research application page on IRAS.
Deferral of registration on a public registry
We recognise that commercial sensitivity may be a concern in some sectors and so it is possible for applicants and sponsors to submit a request to defer registration of clinical trials on a public registry.
Where a deferral is agreed the sponsor is required to publish a minimal record on a public registry. When the deferral period ends, the sponsor should publish the full record on the same registry. When you have done this, email deferrals@hra.nhs.uk to let us know the registration number.
Deferral of research summary on the HRA website
You can also request to defer publication of the HRA Research Summary. This can be included in a request to defer registration or requested separately. If you choose to defer your study, we will still publish a minimum research summary. At the end of the deferral period, we will publish the full research summary.
While a valid deferral is in place we do not expect the summary of results to be made public and the HRA will not publish the lay summary of results, submitted as part of your Final Report, on the research summaries pages.
Deferrals are valid for up to 30 months from the end of the trial, as defined within the clinical trial protocol. Any agreed extensions to the deferral period will extend the previous deferral by another 30 months. The sponsor will be able to defer up to a maximum of 10 years from the end of the trial. After 10 years, the sponsor will not be able to defer registration any further and will have to comply with the transparency requirements. Further deferrals of the registration element 30 months after the end of the study will not normally be agreed unless the sponsor can provide strong justification for doing so. Deferrals after the initial 30 month period will normally only be agreed for publishing the summary of results and, where necessary, sharing results with participants.
Minimum Fields
Where a deferral is agreed, minimal fields will still be published in the research summaries pages, and sponsors are required to register minimal fields on a public registry.
The minimum fields for research summaries of studies with a deferral are:
- REC name
- REC reference
- REC opinion
- date of REC opinion
- IRAS ID
- short title, title of the Research Tissue Bank or title of the Research Database
- study duration (for projects only, not applicable for Research Tissue Banks or Research Databases)
For CTIMPs, where deferral is approved, the minimum information to be published on a registry is:
- registry number
- IRAS ID
- investigator name and location address
- sponsor name and address
- REC decision and date of decision
- nature of clinical trial (for example, bioequivalence in 24 healthy volunteers)
- date of start of trial
- date of end of the trial in the UK, EU member states (if applicable) and globally (if applicable)
- date of start of recruitment
- date of end of recruitment
- justification of deferral
There is more information about registering clinical trials with minimum fields on the ISRCTN Registry website.
How to request a deferral
If you are using Combined Review, you can submit a deferral request in the new part of IRAS, by answering the deferral question in part c of the study information question set.
For other clinical trials not submitted via Combined Review, you can request a deferral by emailing deferrals@hra.nhs.uk with the following information:
- IRAS ID and REC reference
- clear justification for the request
We will confirm whether the deferral has been agreed and provide information about the deferral, including the end date.
After a deferral is agreed: next steps
If we agree a deferral request we will publish a minimum research summary in our research summaries database after the REC final opinion is issued.
If you would like to request an extension to a deferral, you should email deferrals@hra.nhs.uk at least ten working days before the end of the deferral period and include:
- IRAS ID and REC reference
- clear justification for extending the deferral period
We will review requests on a case-by-case basis and will contact you to let you know whether the extension has been agreed and the extended deferral end date.
Any questions?
If you have questions about the deferrals policy or process, email deferrals@hra.nhs.uk
Phase 1 trials
A Phase 1 trial means a clinical trial to study the pharmacology of an investigational medicinal product when administered to humans, where the sponsor and investigator have no knowledge of any evidence that the product has effects likely to be beneficial to the trial participants.
Automatic deferral period
If a sponsor submits a Phase 1 clinical trials of investigational medicinal product (CTIMP) application only involving healthy volunteers from 28 April 2026, it will automatically be deferred for all transparency requirements until 30 months after the end of the trial.
This means a sponsor will not be required to include a written request for a deferral as part of the application. It will be applied automatically upon receipt of a Phase 1 CTIMP only involving healthy volunteers. Confirmation that a deferral is in place will be provided as part of the final approval given to a trial.
Phase 1 healthy volunteer CTIMPs submitted and approved before 28 April 2026 may be given an automatic deferral. However, this depends on whether the end of trial has been declared by this date and whether a deferral is already in place by that point.
The following figure summarises what will happen in each scenario.
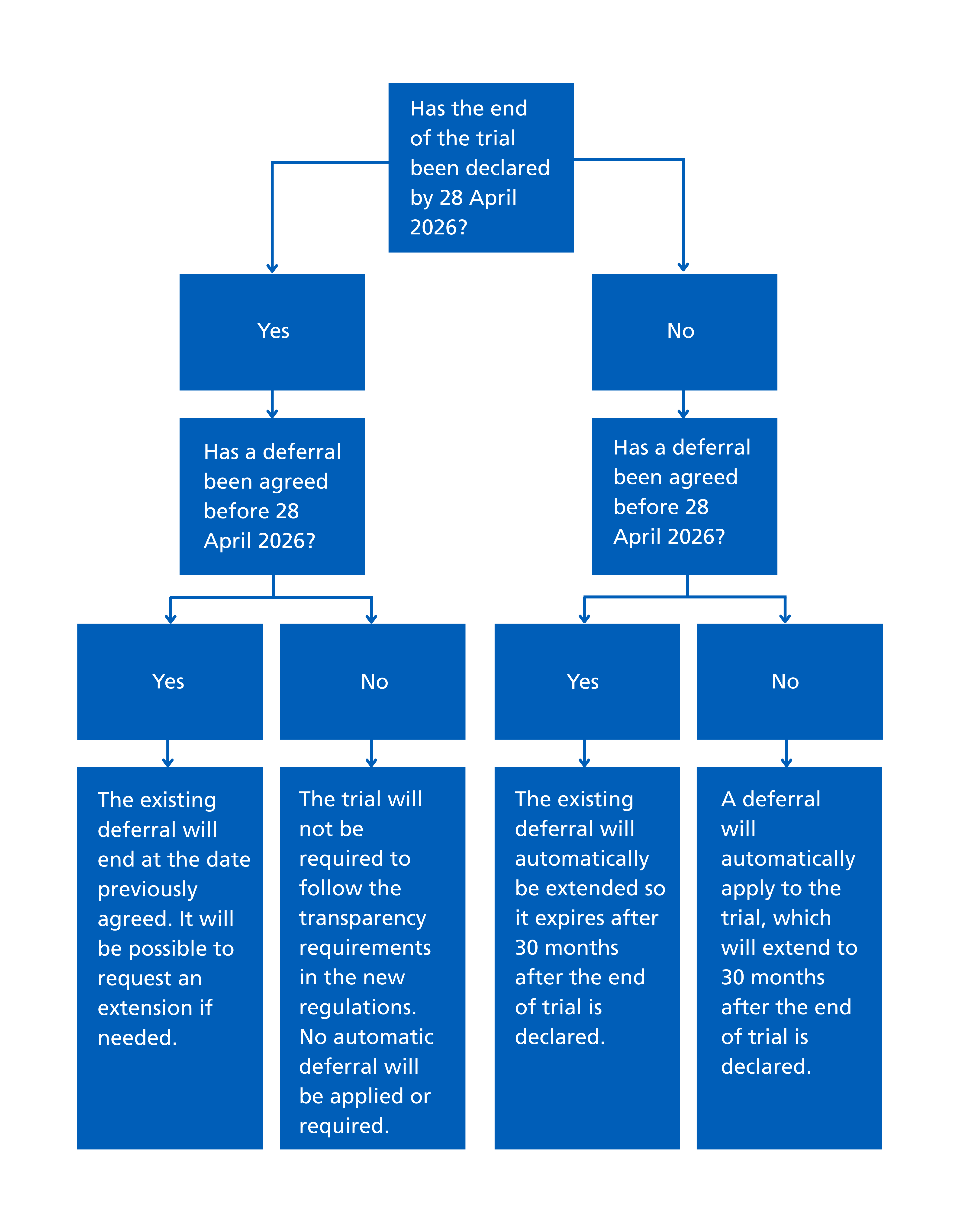
Requirements for Phase 1 trials with automatic deferrals
If a trial has an automatic deferral in place, the sponsor will not have to:
- register the trial in full until 30 months after the trial has ended, however the sponsor must still publish a minimal record on a publicly accessible registry (a minimal record can be published with ISRCTN)
- publish the summary results of the study until 30 months after the trial has ended
- offer to share results with participants during the deferral period
Where a trial qualifies for the automatic deferral, the sponsor will still need to carry out the activity in the 'requirements once a deferral is agreed' section of our deferrals guidance.
It’s still important for sponsors to thank participants or relevant people and provide them with a timeframe for receiving results, even if a deferral is in place.
Although all Phase 1 CTIMPs involving only healthy volunteers submitted from 28 April 2026 will be provided with an automatic deferral, it’s up to the sponsor to decide whether to use this deferral or not.
This means that if the sponsor chooses to register the study, publish summary results, and/or offer to share findings with participants during the automatic deferral period, they can do so.
Extending the deferral for publishing and offering to share summary results with participants beyond the automatic deferral period
We acknowledge that, for Phase 1 healthy volunteer trials, legitimate concerns around commercial confidentiality will often continue beyond the initial automatic deferral period, particularly in relation to the publication of summary results and the offer to share them with participants.
To address these concerns, the sponsor will be able to request, in the cover letter accompanying their initial clinical trial application, 2 additional consecutive 30 month deferral periods beyond the automatic deferral period to cover publication of the summary of results and offering to share results with participants.
Where a sponsor requests these extensions on the grounds of commercial confidentiality, the request will normally be granted unless there are exceptional circumstances. Sponsors should request 2 additional 30 month extensions as part of the initial clinical trial application. After the trial is approved sponsors will normally only be able to extend their automatic deferral by following the process for extending deferrals as outlined in our deferrals guidance.
This will mean that a sponsor of a Phase 1 healthy volunteer trial will not be required to publish the summary of results or offer to share results with participants until 90 months (7.5 years) after the end of the trial.
If the sponsor reaches the end of this 90 month period and wants to defer further, they will be able to request 1 additional 30 month extension by following the process outlined in our deferrals guidance.
Where a sponsor requests this extension on the grounds of commercial confidentiality, the request will normally be granted unless there are exceptional circumstances, for example, if the trial is no longer commercially confidential because summary results have already been made public.
In total, sponsors will be able to defer up to a maximum of 10 years from the end of the trial. After 10 years, the sponsor will have to comply with the transparency requirements.
We do not expect requests for the additional 60 month extension, or any deferral requests extending beyond 30 months after the end of the trial, to include the registration requirement, as these requests will not normally be approved except in exceptional circumstances.
Deferrals for Phase 1 CTIMPs involving patients
If a trial is a Phase 1 CTIMP involving patients it will not automatically be deferred, even if it will involve both healthy volunteers and patients. Sponsors of these trials will be able to request an initial deferral of the transparency requirements by following the process in our deferrals guidance.
Sponsors of phase 1 trials involving patients will also be able to request 2 additional 30 month extensions to the deferral to publish summary results and offering to share results with participants when requesting an initial deferral.
Sponsors should request this as part of their initial clinical trial application. Once a trial is approved, sponsors will normally only be able to extend their initial deferral by following the process described in our deferrals guidance.
If this is requested and agreed, the sponsor will not need to complete these activities until 90 months (7.5 years) after the end of the trial. However, we still recommend that sponsors of Phase 1 trials involving patients thank the participants for taking part in their trial and explain to them, and any other relevant people, at what point they might receive the results of the study.
Waivers for exceptional circumstances
Requesting a waiver from meeting any of the research transparency requirements in the new legislation will be possible.
If a waiver is given, the sponsor will never be required to fulfil the research transparency requirements for the trial.
This differs to a deferral which only postpones the date in which the sponsor needs to meet the transparency requirements.
A request for a waiver should only be made in exceptional circumstances, for example, for reasons of national security or national defence.
Waivers will not be given on the basis of a sponsor’s commercial confidential interests. In these cases, the sponsor should instead request a deferral.
If a sponsor is planning a trial and wants to request a waiver, they should contact deferrals@hra.nhs.uk.
The request must describe why a waiver is requested and provide information on the trial.
Any information in the waiver request that would identify a trial, or any individuals or organisations involved in it, will be kept confidential.
After receiving a request, we’ll consider the information provided, respond to discuss the request further and inform the sponsor of the decision.