Combined Review is a process that researchers use to seek approval for new clinical trials of investigational medicinal products (CTIMPs) and combined medicine and device trials.
Researchers make a single application which goes to both the Research Ethics Committee (REC) and Medicines and Healthcare products Regulatory Agency (MHRA) for review at the same time.
Because the regulatory and ethics reviews are done in parallel, any requests for further information (RFIs) are raised jointly.
A single response to these requests leads to a single decision from both reviews.
Study wide review is usually issued at the same time, but may come later if there are still issues to discuss with the applicant.
Below we share data on the REC part of Combined Review.
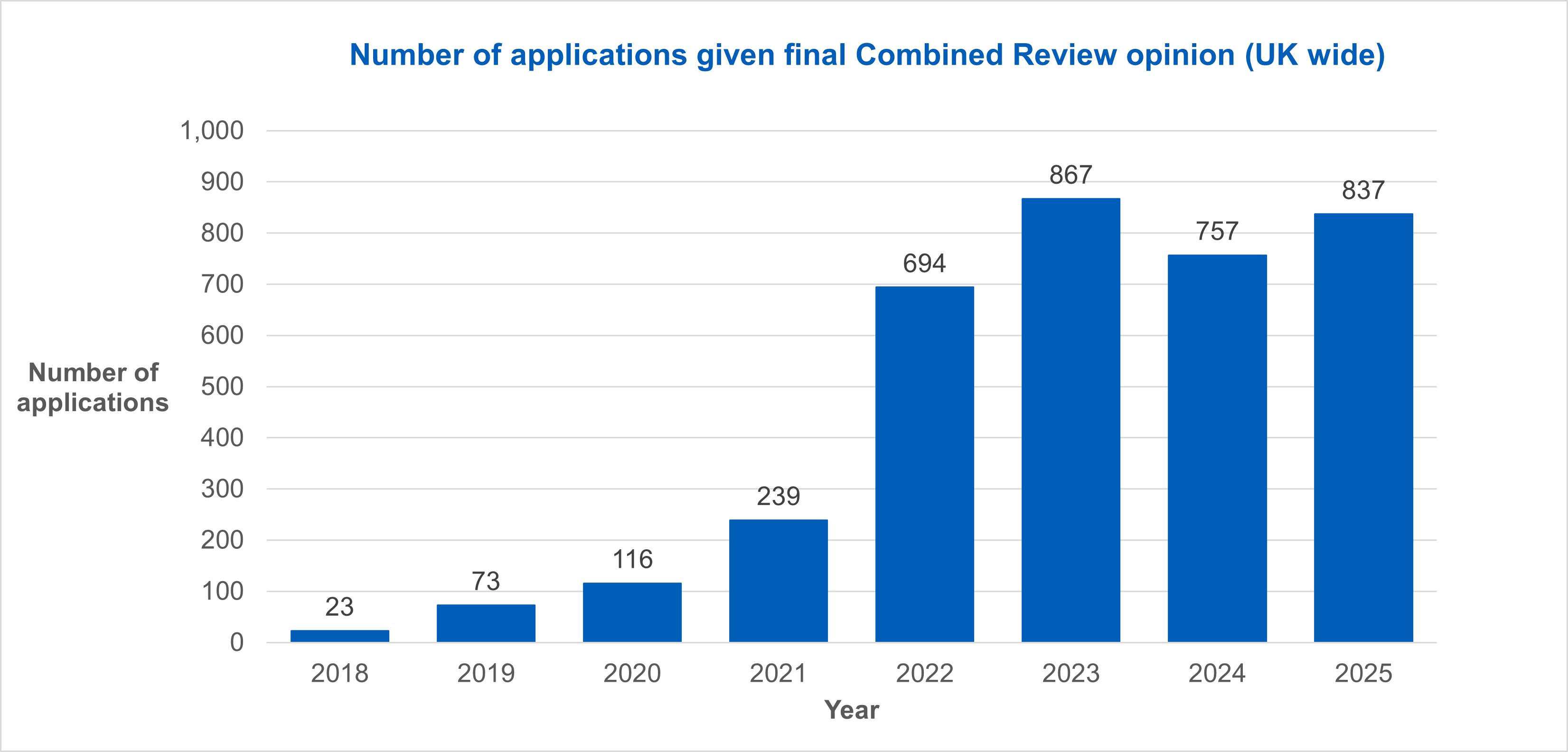
Number of applications given a final outcome via Combined Review
Following a successful pilot, Combined Review was introduced as a requirement for all CTIMP applications in the UK in January 2022.
Since its launch in 2022, we have seen an upward trend in the number of applications given a final opinion via Combined Review.
Review times
Prior to updates made to clinical trials legislation on 28 April 2026 the timeline for the REC part of Combined Review applications was 60 days from the submission of a valid application, unless the REC requested further information from the sponsor before confirming its opinion.
Since using the Combined Review process became a requirement for CTIMP applications in 2022, we have seen a steady improvement in the median number of days taken for a final outcome from a REC to be completed, falling from 37 days in 2022 to 29 days in 2025.
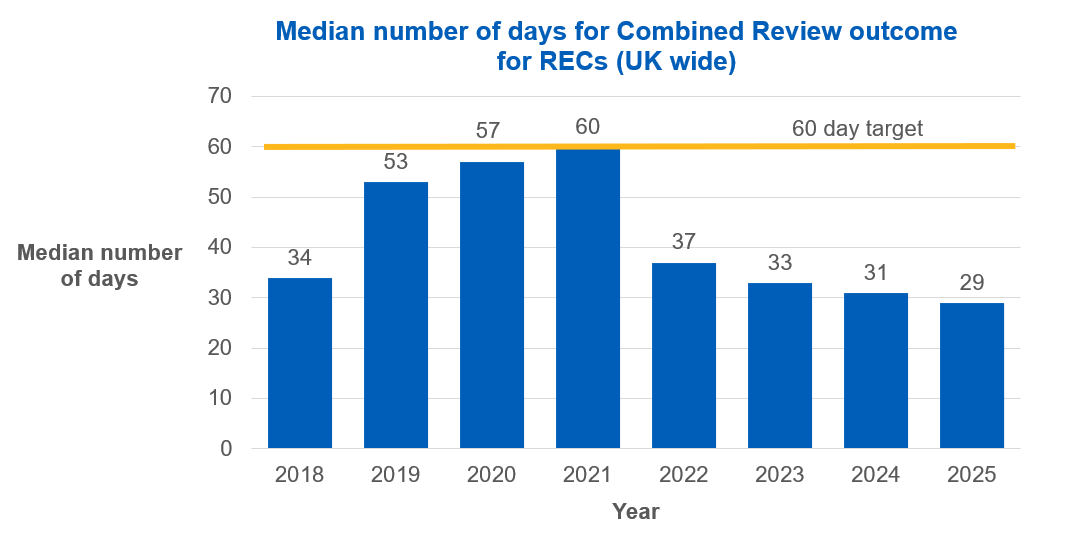
One of the reasons for the fall in the median number of days between 2021 and 2022 is a change in how REC timelines were measured. Prior to 2022 the clock used to measure the time taken was stopped when the outcome was shared with the applicant. However, this method did not accurately measure the actual time being taken by the REC, as the final outcome through Combined Review is shared by the MHRA. From 2022 the clock for the REC was stopped when the committee had made a final decision.
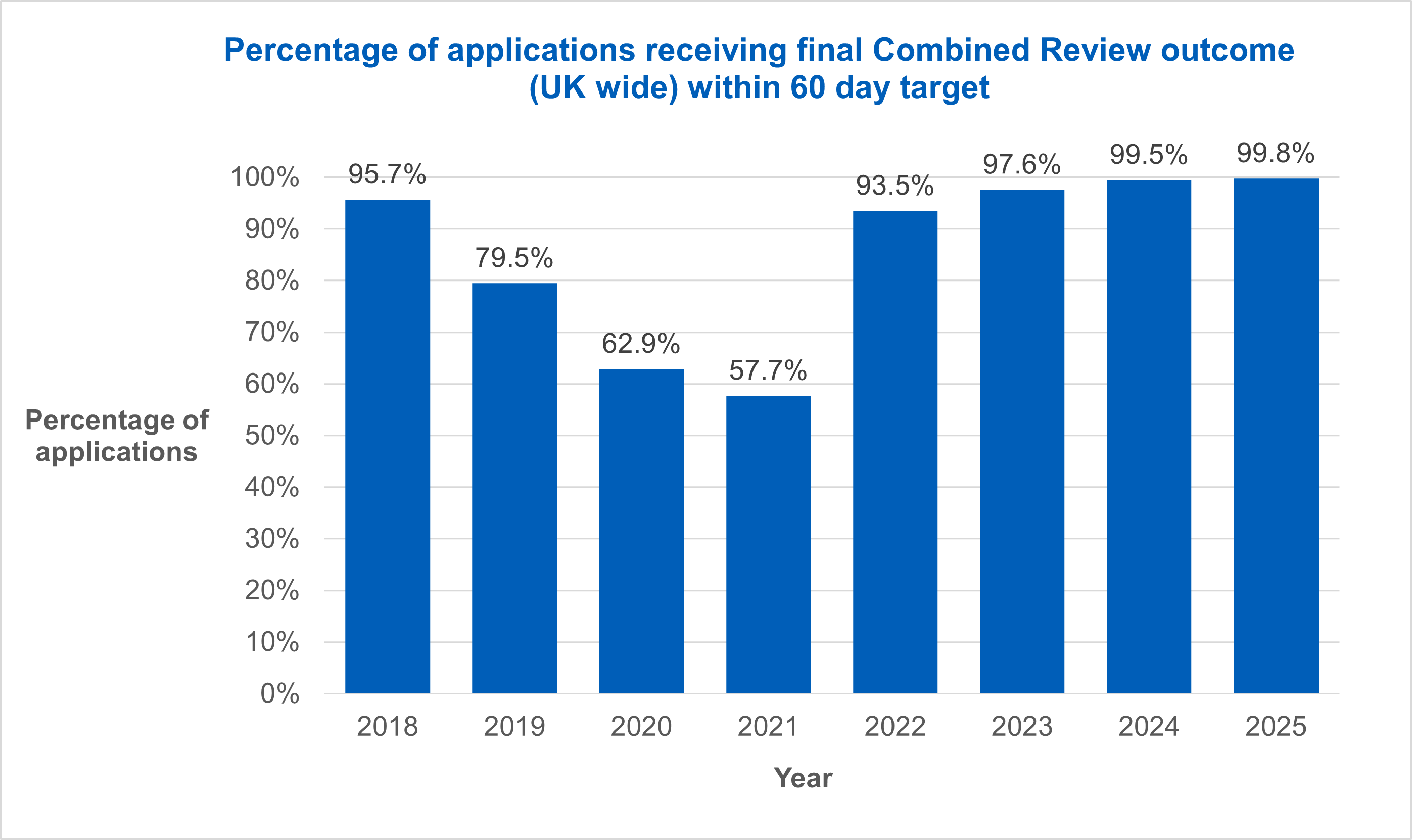
In 2023 the HRA and MHRA were set a target of 99% of studies receiving a final Combined Review outcome within 60 days as part of the key performance indicators that we report on to the Department of Health and Social Care.
When measuring the HRA’s performance against this target, we look at studies that were reviewed by RECs across the UK. The HRA is the appointing authority for RECs in England, and we work closely with Scotland, Wales and Northern Ireland to support a single UK wide ethics service.
From 2022 onwards we have seen a continued improvement in the percentage of applications given a final outcome within the 60 day target.
Over the past two years RECs across the UK have met the 60 day target with 99.5% and 99.8% of final outcomes being completed within the target in 2024 and 2025 respectively.
Latest review times
Below is the monthly median number of days taken for a REC final opinion to be completed through Combined Review.
This data represents the REC review across the UK with figures from England, Wales, Scotland and Northern Ireland:
- January 2026 – 28 days
- February 2026 – 33 days
- March 2026 – 29 days
Monthly breakdown of Combined Review performance
You can take a look at a monthly breakdown of Combined Review performance as part of the UK Clinical Research Delivery key performance indicators.
The Combined Review data shows the REC and MHRA performance data together as ‘indicator number 2’.
The way that we measure Combined Review is changing
When the clinical trials regulations were updated in April 2026 it changed the way that we report on our performance around Combined Review.