On Tuesday 28 April 2026 new clinical trials regulations came into effect, representing the largest overhaul of the law governing Clinical Trials of Investigational Medicinal Products (CTIMPs) in the UK for nearly two decades.
As part of the updated regulations, we’re changing the way that we report our data, so that it better shows the time taken to review applications, and the time experienced by the sponsor.
This means that for a short time, the total timeline for studies from submission to decision may look longer than you are used to seeing reported.
This doesn’t impact our commitment to keep working to make the whole process faster, and we are continuing to make improvements to help speed up the set-up process.
What’s changed?
From Tuesday 28 April 2026 the timeline for Combined Review has changed.
Under the previous regulations, the time taken for applicants to respond to requests for further information (RFI) from the REC were not included in the overall timeline.
If the REC needed more information to make a decision, the applicant had an unlimited amount of time to respond. This period was also known as a ‘clock stop’.
For the MHRA side of Combined Review, the sponsor had 14 days to reply.
Though the maximum number of days for a Combined Review outcome was set at 60 days, the unlimited clock stops (during the REC’s RFI response period) meant that the process could take longer if the applicant needed more time to provide the necessary information, but you would not be able to see this in the reported figures.
The new regulations introduce a maximum 107 day timeline, which includes set time limits for sponsors to respond to an RFI – meaning there are no longer any clock stops.
These changes will provide a more accurate representation of actual approval times as they are experienced, and will help us better understand where there may be challenges at different stages of the process so that we can address them.
Whilst this may at first look like the time available for Combined Review has been extended, 107 days now represents the maximum number of days that are available for the entire process, when before there was no overall limit on the REC’s RFI response period.
In the diagram below we set out the four different stages of the 107 day timeline.
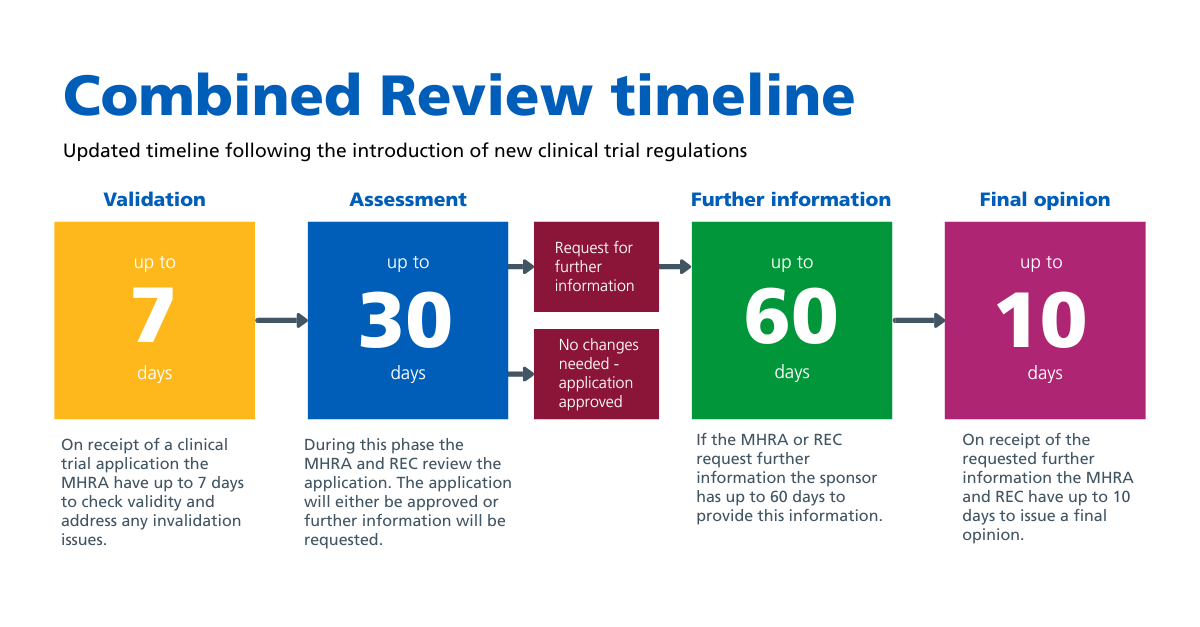
As part of the new timeline we will continue to measure our performance on the phases of the review process that are within our control.
These are the 30 day assessment and the 10 day assessment of an RFI response.
We will not be reporting on the 7 day validation period which is led by the MHRA, or the 60 day RFI phase which will be the responsibility of the research sponsor.
Does this mean Combined Review will take longer?
Based on our performance over the past few years, we expect the time taken for a final outcome from the REC to be completed via Combined Review to remain efficient.
Our existing processes for reviewing CTIMP applications via Combined Review, which have been performing strongly for a number of years, remain unchanged.
Our strategy for 2025-28 outlines our aim for 100% of Combined Review final outcomes to be completed within 60 days, with a median timeline below 40 days. This does not include the time taken for the validation of an application or for the sponsor to respond to any RFI.
In 2025 the median time for final outcomes to be completed was 29 days (RECs UK wide).
Our ongoing work as part of the UK Clinical Research Delivery (UKCRD) programme aims to speed up the time taken for Combined Review even further.