Our review and approval of applications for health and social care research in the UK is happening faster than ever according to a review of data from 2025.
We've taken a look back at our performance data from 2025 which showed that the total number of applications reviewed by Research Ethics Committees (REC) in the UK increased by 3.1%, and the time taken for these reviews to be carried out reduced to 29 days for commercially sponsored studies in 2025 – less than half of the target of 60 days.
99.8% of reviews of Clinical Trials of Investigational Medicinal Products (CTIMPs) in the UK were completed by the REC within the 60 day target under Combined Review, with this taking a median of 29 days.
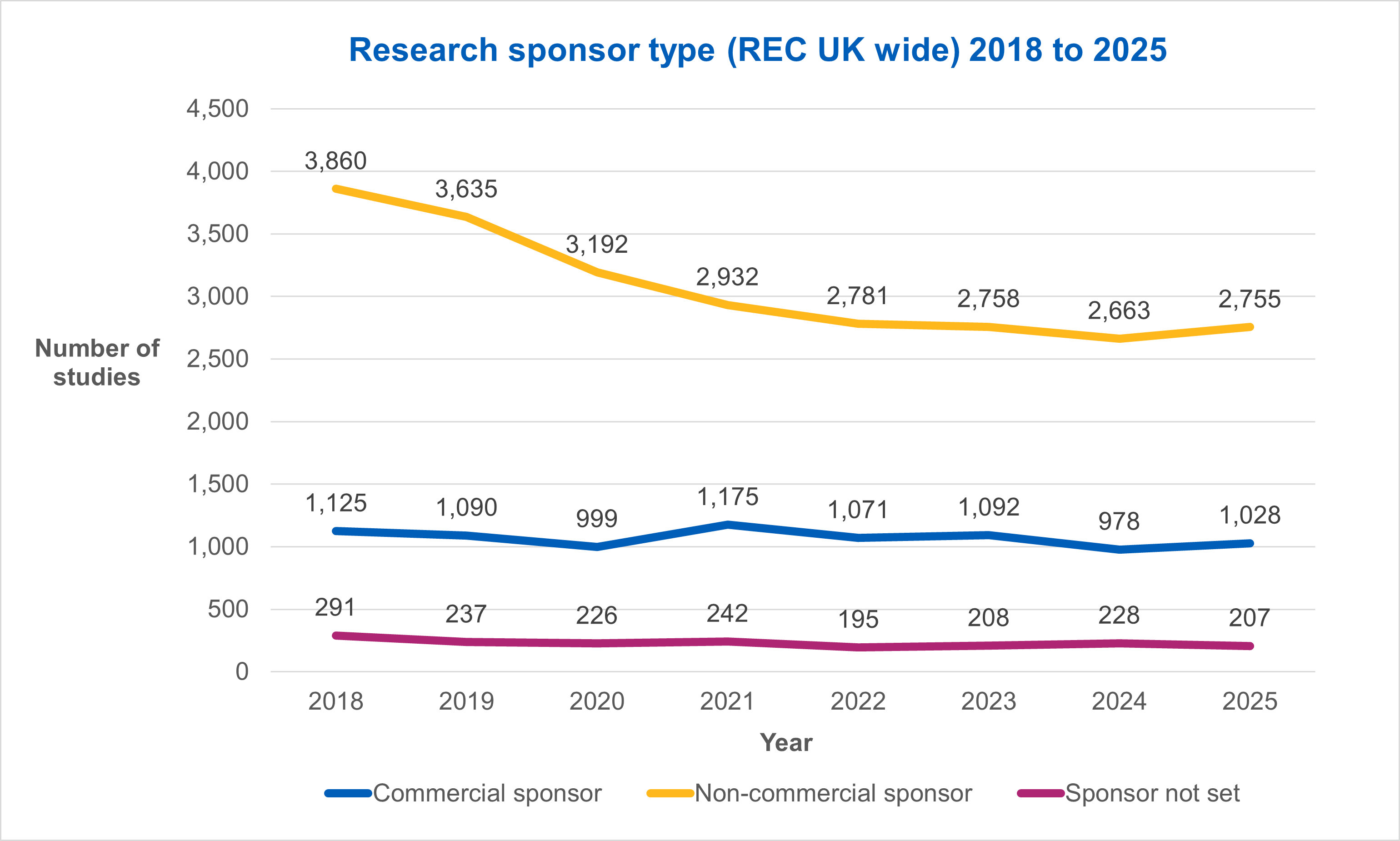
Applications for commercially sponsored studies rose by 5.1%, from 978 in 2024, to 1,028 in 2025. Similarly, applications for non-commercially sponsored studies rose by 3.4%, from 2,663 in 2024, to 2,755 in 2025.
The number of technical assurances (specialist pre-application reviews) provided by the HRA also increased in 2025, with a record number of Pharmacy Assurances (171) given during the year. The time taken for Pharmacy Assurance to be provided was also at a record low, taking just 22 days.
Summary of our findings
- in 2025 99.8% of CTIMPs reviewed under Combined Review by a REC in the UK were completed within the 60 day target
- the number of studies receiving a final opinion from a REC (UK wide) rose by 3.1% from 3,869 in 2024, to 3,990 in 2025
- the number of studies receiving HRA and Health and Care Research Wales (HCRW) Approval also rose by 3.1% from 3,408 in 2024, to 3,515 in 2025.
- 130 applications received a final outcome from the Confidentiality Advisory Group, up from 120 in 2024
- 7,727 substantial amendments were reviewed, a fall of 1.9% from 7,880 in 2024
- 13,680 non-substantial amendments were reviewed, up 2.2% from 13,386 in 2024
- the number of applications for commercially sponsored studies rose by 5.1%, from 978 in 2024, to 1,028 in 2025
- 12.3% increase in the number of studies reviewed by a REC (UK wide) under the Proportionate Review process
- a record number (173) of Pharmacy Assurances were provided in 2025
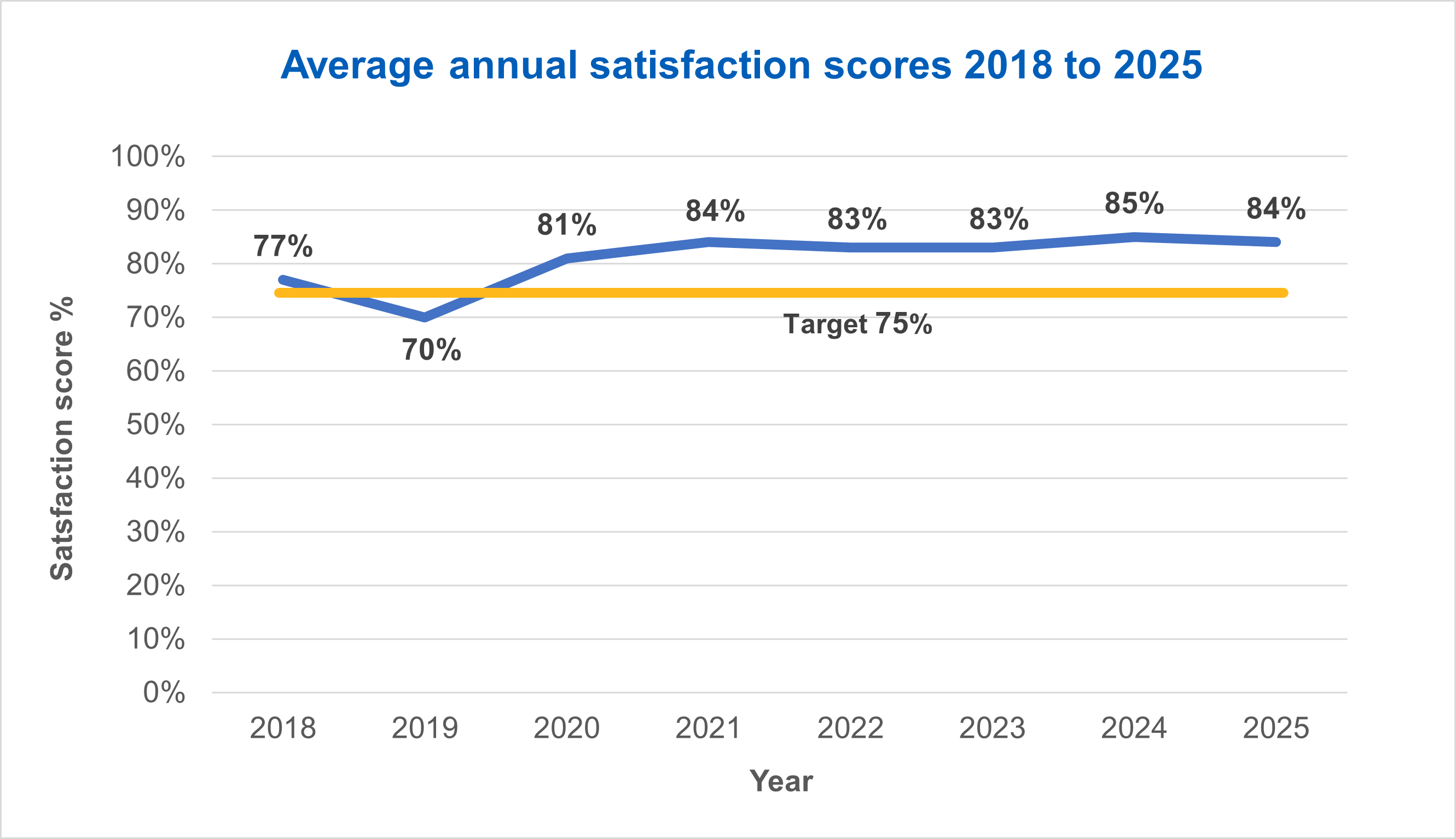
Satisfaction
Satisfaction remained high, with 84% of people who gave feedback on working with the HRA rating their experience at least a 7 out of 10 or higher.
For the sixth year in a row the HRA exceeded its target of at least 75% satisfaction.
Combined Review
Combined Review is a single application for the review of a Clinical Trial of Investigational Medicinal Products (CTIMPs) that is reviewed by a Research Ethics Committee and the Medicines and Healthcare products Regulatory Agency (MHRA) at the same time.
As part of the review every application is reviewed by a REC based in England, Wales, Scotland or Northern Ireland.
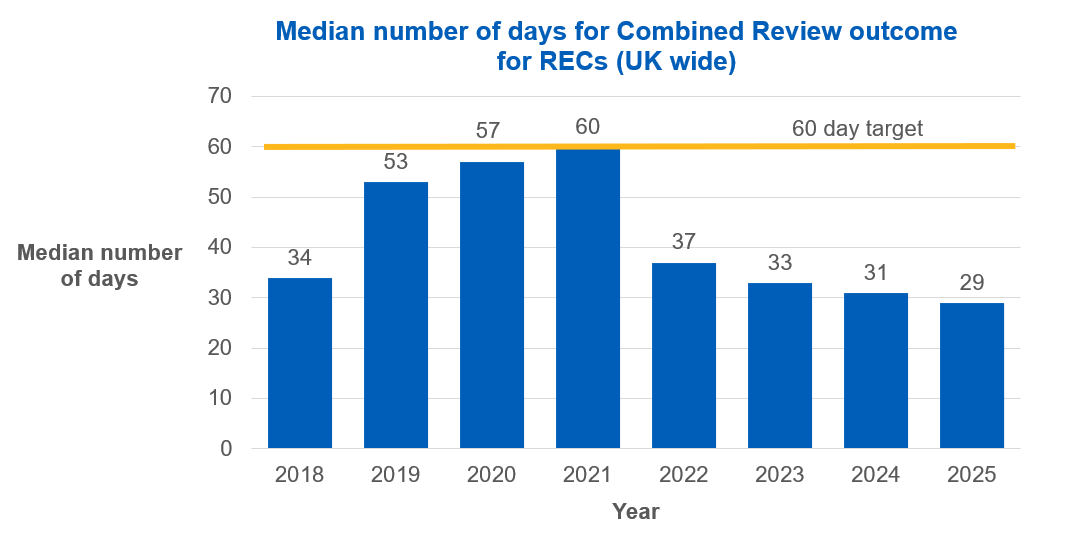
837 applications were given a final opinion (UK wide) via Combined Review in 2025. The median number of days taken for a Combined Review outcome to be completed (UK wide) was 29 days, less than half of the 60 day target. This is well below our 40 day target outlined in our strategy for 2025-2028.
In 2025, 99.8% of applications reviewed under Combined Review by RECs across the UK had a completed final outcome within the 60 day target.
Review times
Depending on the type of study an application may be reviewed by a Research Ethics Committee, reviewed for HRA and HCRW Approval, or both.
In 2025 the time taken for the HRA to review studies continued to perform strongly:
- amendments – the time taken for substantial amendments to be reviewed by a REC (UK wide) was 19 days and for HRA and HCRW Approval was 28 days, well below our 35 day target for the sixth consecutive year
- Confidentiality Advisory Group – for the eighth consecutive year the time taken for final outcomes were well below our target of 30 days for precedent set studies and 60 days for studies requiring a full review
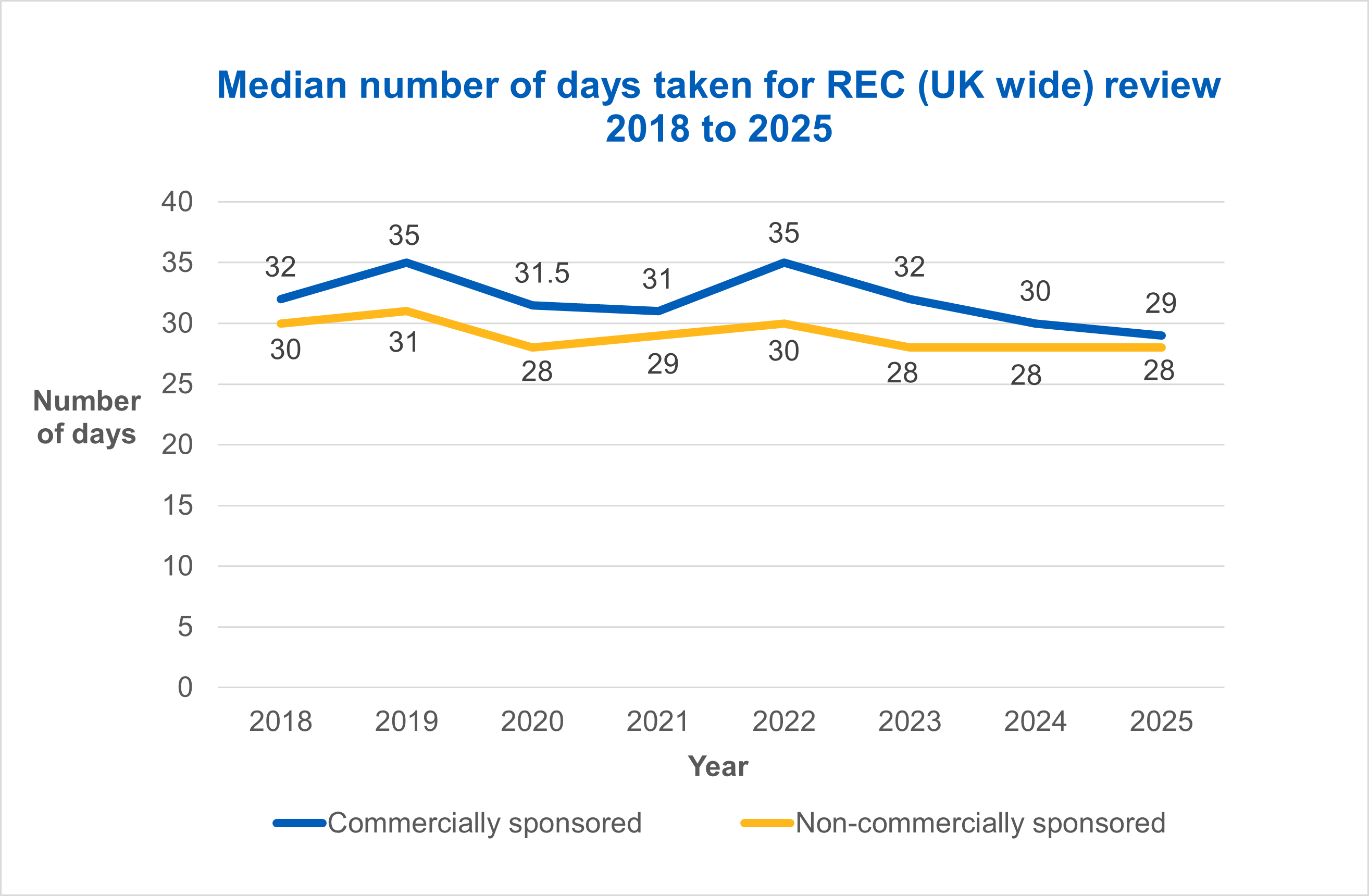
- HRA and HCRW Approval – the median days taken for commercially sponsored studies dropped from 74 days in 2024, to 73 days in 2025, but increased slightly for non-commercially sponsored studies from 68 days in 2024 to 70 days in 2025
- REC (UK wide) final opinion – the median days taken for a final opinion for commercially sponsored studies dropped from 30 days in 2024 to 29 days in 2025, and for non-commercially sponsored studies remained at 28 days - well under the 60 day target
- Proportionate Review – the time taken for studies to receive a REC final opinion (20 days) or HRA and HCRW Approval (45 days) increased slightly
- Pharmacy Assurance – the median days taken was 22 days, well below the 30 day target
- Radiation Assurance – the median days taken was 41 days, just above the 40 day target
Number of studies reviewed
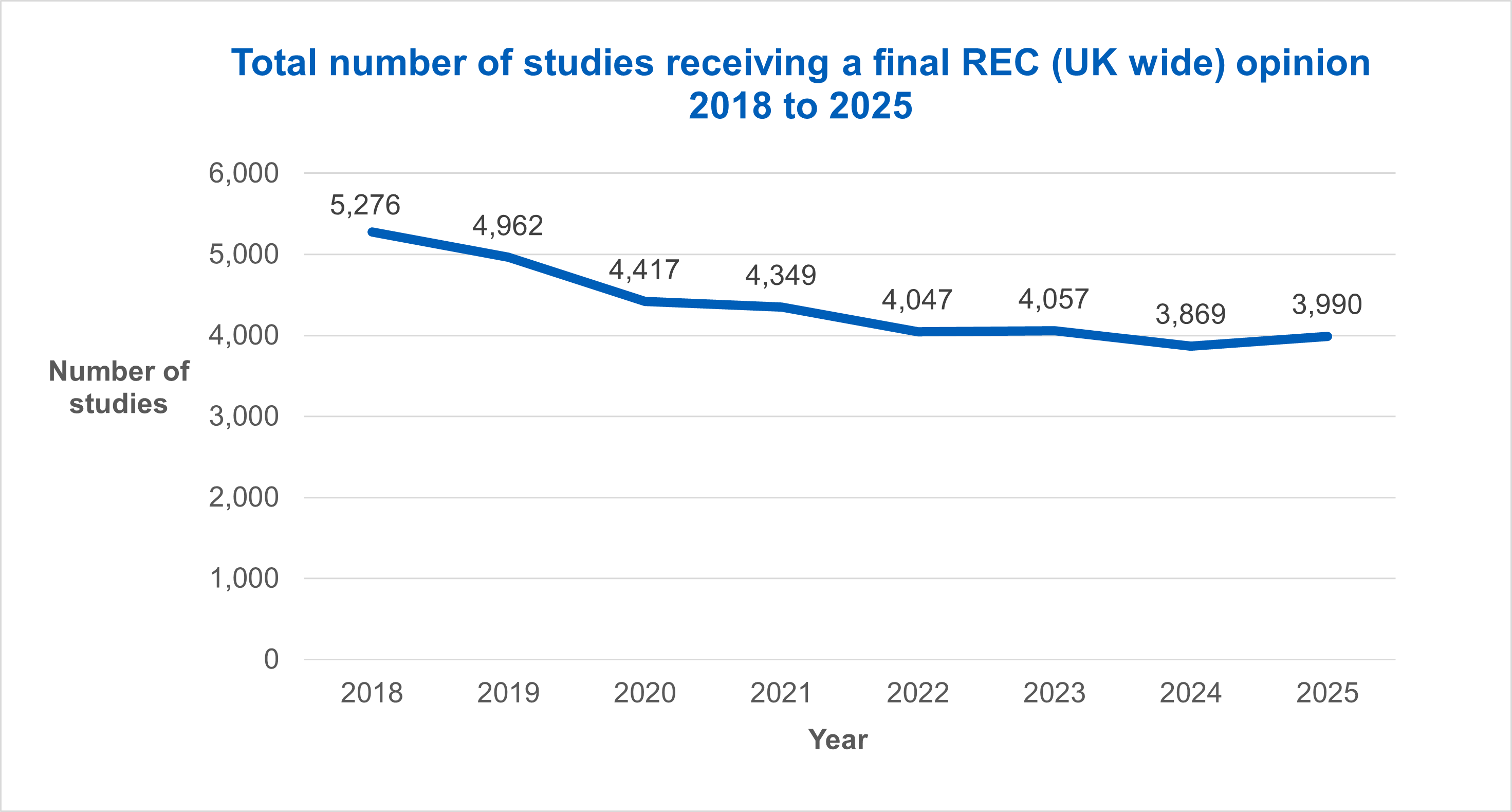
The total number of applications receiving a final outcome from a Research Ethics Committee (UK wide) rose by 3.1% from 3,869 in 2024, to 3,990 in 2025.
This trend was mirrored in the number of applications receiving HRA and HCRW Approval, which also rose by 3.1% from 3,408 in 2024, to 3,515 in 2025.
Within these trends we saw a 5.1% increase in the number of commercially sponsored studies that received REC review, and a 3.4% increase in non-commercially sponsored studies.
Research sponsor type
We saw a rise in the number of applications from commercial and non-commercial sponsors.
Commercial sponsor applications rose from 978 in 2024, to 1,028 in 2025.
Similarly, non-commercial sponsor applications rose from 2,663 in 2024, to 2,755 in 2025.
Our response to the data from 2025
Responding to the data Jonathan Fennelly-Barnwell, Interim Director of Approvals at the Health Research Authority, said:
Jonathan Fennelly-Barnwell, Interim Director of Approvals“In 2025 we launched an ambitious new strategy where we outlined how we would continue our work to make it easier and faster to do research in the UK.
“Over the past 12 months as part of our work with partners on the UK Clinical Research Delivery (UKCRD) programme we have introduced a range of changes designed to help streamline the application process.
“We have further streamlined the commercial contracting process to help remove unnecessary duplication when setting up studies, and improved consistency around the use of technical assurances.
“Despite an increase in the number of applications submitted, our data shows that we have been able to maintain our review times, and in some areas speed them up. This was one of the key priorities set for us by the government as part of their target to speed up the set-up time for studies to 150 days by the end of March 2026.
“We know how important speed is for researchers when it comes to the approvals process, but we also know that it is just as important for the processes to be reliable and predictable so sponsors can plan their studies effectively.
“What this data shows is that the UK is a fast, reliable place to carry out research, where there are consistent timelines that researchers can confidently plan their research around.
“We’re proud to see the impact of the improvements we are making across the board, but of course there’s still lots more work to be done.
“The new clinical trials legislation coming into effect today will help support our work to speed up processes. The reforms will help better align processes for Combined Review and modifications to applications, and make it easier to start lower-risk studies.
“Our work on Plan and Manage Health and Care Research will also help by creating a new seamless digital service that will make it easier complete every step of the research application process from start to finish.
“In December 2025 we launched the first service with a small group of sponsors which focuses on amendments. We will expand this service to bring more sponsors on board during 2026. Whilst we are confident that this will help speed up the amendment process long term, we know that in the short term such a big change may impact on timelines. We will support sponsors throughout the process to make the transition as easy as possible.
“None of what we do as an organisation would be possible without the amazing contribution of our volunteers. We are lucky to work with nearly 1,000 volunteers who give their valuable time and expertise to sit on Research Ethics Committees or our Confidentiality Advisory Group. I want to take this opportunity to say thank you. We couldn’t do what we do without you.”
Other key findings
Amendments
Amendments are changes proposed to a research project after it has already been approved.
We saw a 1.9% fall in the number of substantial amendments and a 2.2% increase in the number of non-substantial amendments in 2025.
The total number of combined substantial and non-substantial amendments was relatively unchanged, increasing slightly from 21,266 in 2024, to 21,407 in 2025.
The median number of days for a REC (UK wide) to review an amendment remained at 19 days, which is unchanged since 2020 and well below our 35 day target.
CAG
In 2025 we saw a small increase in number of studies given final outcome by CAG, up from 120 in 2024 to 130 in 2025.
In 2025 for a full review carried out by CAG we saw a slight increase in the median number of days taken. Full reviews took 31 days (research) and 25 days (non-research), up from 30 days and 24 days in 2024 respectively.
For precedent set reviews we saw a slight decrease in the time taken for a review to be completed. Precedent set reviews took 23 days (research) and 15 days (non-research), down from 24 days and 17 days in 2024 respectively.
Technical Assurance
Technical Assurance is a pre-submission review process that we offer for studies that involve pharmacy (CTIMPs) or ionising radiation (which includes things like x-rays and radiotherapy).
A record number (171) of Pharmacy Assurances were provided during 2025, up 23% from 139 in 2024.
The median number of days for Pharmacy Assurance to be provided in 2025 was 22 days, down from 23 days in 2024, and well below our 30 day target.
We also saw a rise in the number of Radiation Assurances provided (173) in 2025, up from 163 in 2024.
The median number of days for Radiation Assurance to be provided reduced from 47 days in 2024, to 41 days in 2025. This is still one day above our 40 day target, but a significant improvement on the 53 day record high in 2022.
Our performance data in more detail
We have published a more detailed analysis of our performance data between 2018 and 2025.
You can find a summary of our performance during this period, alongside the latest data for 2025 in the following areas:
The way we measure and report on Combined Review is changing
On Tuesday 28 April 2026 new clinical trials regulations came into effect, representing the largest overhaul of the law governing Clinical Trials of Investigational Medicinal Products (CTIMPs) in the UK for nearly two decades.
As part of the updated regulations, we’re changing the way that we report our data, so that it better shows the time taken to review applications, and the time experienced by the sponsor.
This means that for a short time, the total timeline for studies from submission to decision may look longer than you are used to seeing reported.
This doesn’t impact our commitment to keep working to make the whole process faster, and we are continuing to make improvements to help speed up the set-up process.